Resolution of Enantiomers
| Home | | Organic Chemistry |Chapter: Organic Chemistry : Stereochemical and Conformational Isomerism
We have seen that individual enantiomers have identical physical properties and only can be distinguished in a chiral environment. Plane-polarized light is such a chiral environment, and one enantiomer is dextrorotatory and one is levorotatory.
RESOLUTION OF ENANTIOMERS
We
have seen that individual enantiomers have identical physical properties and
only can be distinguished in a chiral environment. Plane-polarized light is
such a chiral environment, and one enantiomer is dextrorotatory and one is
levorotatory. Another way to distinguish enantiomers is to allow them to react
(or interact) with other chiral molecules. The interaction of a mixture of
enantiomers with a single enantiomer of a chiral molecule produces a mixture of
diastereomers as illustrated.
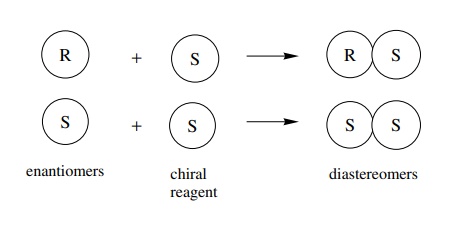
Since
diastereomers have different physical properties, they can be separated on the
basis of those physical properties. After separation of the diastereomers, the
individual enantiomers are reclaimed, and in this way the two enantiomers will
have been separated. Such interactions form the basis for all separations of
racemic mixtures into pure enantiomers, which is termed “resolution” of
enan-tiomers. There are several experimental techniques used to resolve
enantiomers, but all utilize a chiral reagent of some type to furnish the
chiral environment needed to distinguish the enantiomers.
Crystallization
has been a traditional method for separating the diastereomers produced from a
racemic mixture and a chiral resolving agent. For example, racemic carboxylic
acids can be treated with an optically active alkaloid (which is basic) and the
resulting diastereomeric salts are separated by crystallization. The individual
enantiomeric acids are then regenerated from the salts. A vari-ety of alkaloids
have been used as resolving agents for racemic acids. They include brucine
strychnine, ephedrine, quinine, morphine, and α-phenyl ethy-lamine, among others.
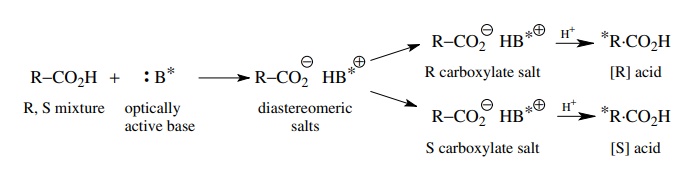
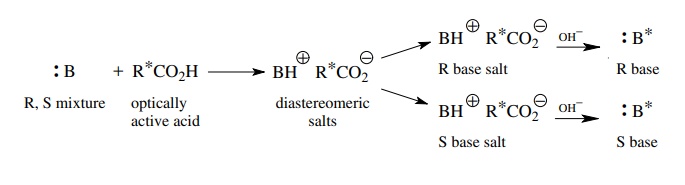
Racemic
bases can be resolved by treating them with an optically active acid and
separating the resulting diastereomeric salts by fractional crystalliza-tion.
The individual enantiomeric bases are regenerated from the salts. Common
acid-resolving agents include camphorsulfonic acid and derivatives of it,
tartaric acid, malic acid, and pyroglutamic acid, among others.
Alcohols
are often resolved by conversion to half esters of phthalic acid or succinic
acid, which are then resolved as typical acids. The alcohol is then
regen-erated from the resolved half ester by hydrolysis or reductive cleavage
with LAH. A second method for resolving alcohols is to convert them to esters
of optically active acids. This gives a mixture of diastereomeric esters which
are separated by fractional crystallization and the alcohol is recovered by
hydrolysis or reductive cleavage.
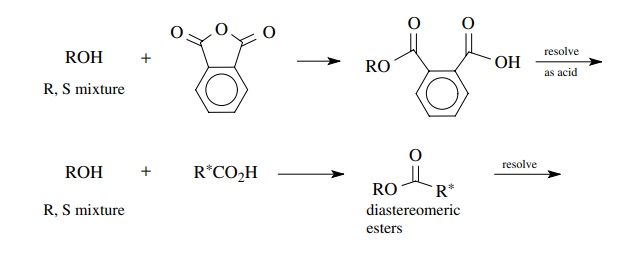
In
recent times chromatography has become a major technique for separations and
has increasingly supplanted fractional crystallization as a way to separate
diastereomeric compounds. Not only do diastereomers have different
solubilities, they also interact with surfaces such as silica gel or alumina
differently. The mix-ture of diastereomeric esters obtained by coupling a
racemic alcohol to optically active acid can often be separated by
high-performance liquid chromatography (HPLC), radial chromatography, or flash
chromatography. Chromatography is often much faster and more efficient than
crystallization. The individual alcohols can be regenerated in the usual
fashion.
The
preceding methods for the resolution of enantiomers rely on the forma-tion of
strongly bound diastereomers (ionic or covalent) which are then sep-arated. It
has become more and more common to use weak interactions as a means of
resolving enantiomers. Chiral chromatography columns are useful for the
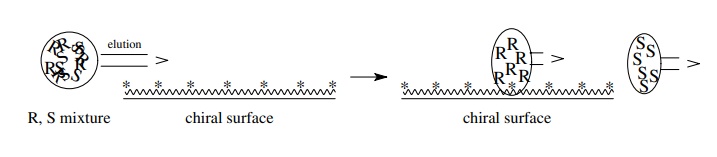
separation of a variety of compounds, including amino acids. A chi-ral substance
is permanently attached to the column surface. If a mixture of enantiomers is
passed over the surface, the individual enantiomers will interact with the
chiral surface differently and thus will elute along the column at dif-ferent
rates. (The enantiomer which interacts with the surface more strongly will
elute more slowly). They can thus be collected individually. A variety of
chiral stationary phases are available to separate an ever increasing number of
examples.
The
use of enzymes to resolve enantiomers has become an extremely popular method
only recently. Enzymes are chiral catalysts which often exhibit very high
selectivity for one enantiomer of a racemic mixture. Since enzymes are soluble
in aqueous solution, it was often impossible to get sufficiently high
concentrations of organic substrates in the aqueous medium to achieve
conversion at any reasonable rate. The finding that a variety of esterases
(lipases) can function very well in organic solvents has removed this major
stumbling block to the practical utilization of biochemical transformations for
resolution. In addition, a variety of enzymes are available commercially.
Moreover a variety of other enzymes are now available and can be used to
resolve enantiomers effectively. For example, some amidases and peptidases
(amide bond hydrolysis) can be applied to the resolution of enantiomers.
Thus
it is very easy to acetylate a racemic alcohol and treat the racemic mixture of
acetates with a lipase. One enantiomer is hydrolyzed to the alcohol and the
other remains as the ester. These are separated chromatographically and each
component is obtained with high optical purity. This technique is becoming more
important and could be the most general technique for resolution in the future.

The
use of kinetic resolution to obtain a single enantiomer from a mixture of
enantiomers is often useful for particular functional groups. Since individual
enantiomers react at different rates with chiral reagents, treatment of a
racemic mixture with a limited amount (0.5 equiv.) of a chiral reagent will
convert one of the enantiomers to product in preference to the other. After
workup one enan-tiomer will be recovered unchanged while the other will have
been converted to a new product. The efficiency of the kinetic resolution will
depend on the relative rates of reaction of the two enantiomers. If rates of
reaction (selectivity factor) vary by >100,
then the recovered enantiomer will be >99%
optically pure. Lower selectivity factors will lead to less pure enantiomers.
Optically
active diisopinocamphenylborane can be used to resolve racemic olefins. The
reagent adds to one enantiomer, and the other is unchanged. Opti-cal purities
on the order of ∼37 – 65% are
possible. Chiral allylic alcohols can be resolved with chiral epoxidizing
agents derived from tartrate complexes of titanium. One enantiomer is
epoxidized and the other is not. Thus, the two alco-hol enantiomers can be
separated, one as the unsaturated alcohol and one as the epoxy alcohol. Use of
the other tartrate isomer reverses the stereoselectiv-ity. Selectivities on the
order of >100 are possible with
this method. As in any kinetic resolution, however, only one enantiomer can be
recovered. The other is converted to a different chiral product.
Even
if the separation of enantiomers by any of the above methods is not completely
successful, it is often possible to further raise the enantiomeric excess by
crystallization or chromatography. In this way many pure enantiomers are now
available.
Related Topics
