Robinson Annulation
| Home | | Organic Chemistry |Chapter: Organic Chemistry : Planning Organic Syntheses
Another example of a very common ring-forming sequence is the Robinson annulation. This sequence allows a six-membered ring to be appended to an existing carbonyl group.
ROBINSON ANNULATION
Another example of a very common ring-forming sequence is the Robinson annulation. This sequence allows a six-membered ring to be appended to an existing carbonyl group.
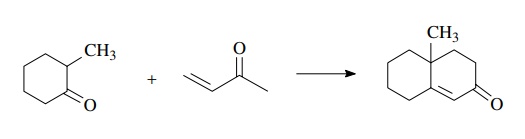
The
strategy of the sequence is a Michael addition to an α,β-unsaturated ketone
followed by an intramolecular aldol reaction. Treatment of a ketone enolate
with a Michael acceptor gives a diketone intermediate which is poised to
produce a six-membered ring if an enolate is produced and it intramolecularly
adds to the carbonyl group.
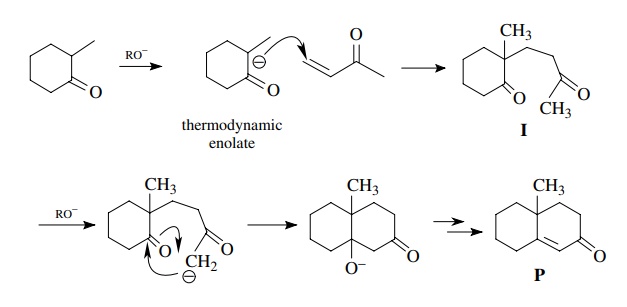
This
process nicely accounts for formation of product, but if we consider
intermediate I, we see that there
are several different α protons that
could be removed by base, Ha
, Hb , and Hc . Furthermore the acidities
of these various α protons should be
comparable so all should be removed to similar extents under the reaction
conditions. If we sequentially remove each proton and write the product from an
intramolecular carbonyl addition, the following products could be produced. The
fact is that only P is produced to
any extent. This is due to the preference of six-membered ring formation over
the formation of the more strained four-membered ring product or the more
strained bridged product. Thus the enolate formed by the removal of Ha closes faster than the
enolates formed by removal of Hb
or Hc .
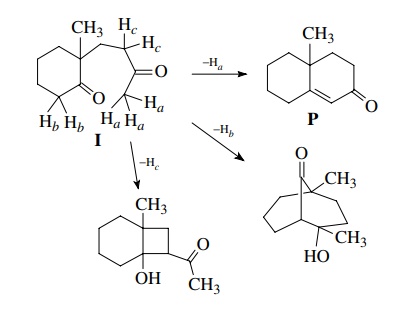
Furthermore,
since the aldol reaction is reversible, if any of these higher energy products
were formed, they could open back up under the reaction conditions. The
exclusive formation of P is an
example of kinetic as well as thermodynamic control as the more stable product
is formed fastest.
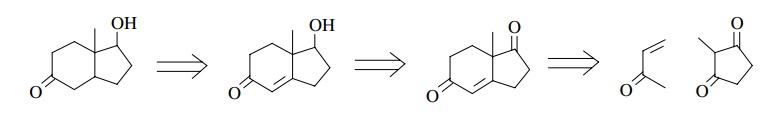
For
the purposes of retrosynthetic analysis, a six-membered ring in a target can be
related to a Robinson annulation of an existing ketone with an α,β-unsaturated
ketone. Normally α,β-unsaturated methyl ketones are used to
facilitate the ring closure, but this is not an absolute requirement. Thus the
target steroid S could potentially
be constructed by a series of Robinson annulations as shown. The last
retrosynthetic step (the first synthetic step) could be problematic as a
mixture of regioisomers would be formed.
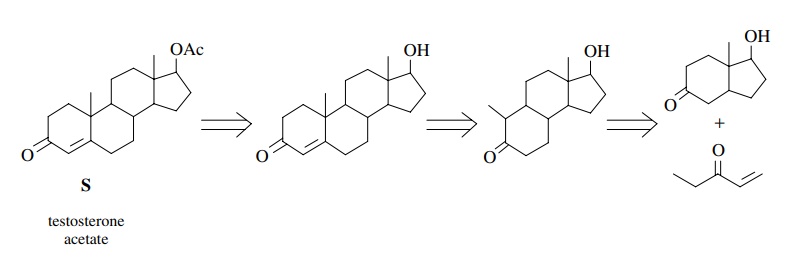
Furthermore
the bicyclic starting material could also be constructed by a Robinson
annulation on a cyclopentanedione. In this case the final functionality must be
achieved by selective reductions of the olefin and ketone functions at
appropriate stages in the synthesis.
A
great many complex multicyclic targets have been synthesized by the Robinson
annulation attesting to its generality and versatility.
Related Topics
