Spin-Spin Coupling
| Home | | Organic Chemistry |Chapter: Organic Chemistry : Structure Determination of Organic Compounds
The determination of the numbers of different kinds protons in a molecule is a very important use of NMR spectroscopy, but it does not establish the connectivity of the carbons bearing those protons and the connectivity is crucial in correct structure determination.
SPIN-SPIN COUPLING
The
determination of the numbers of different kinds protons in a molecule is a very
important use of NMR spectroscopy, but it does not establish the connectivity
of the carbons bearing those protons and the connectivity is crucial in correct
structure determination. However, NMR spectroscopy can also give insight into
the connections between functional groups by the presence of spin – spin
coupling in the NMR spectrum.
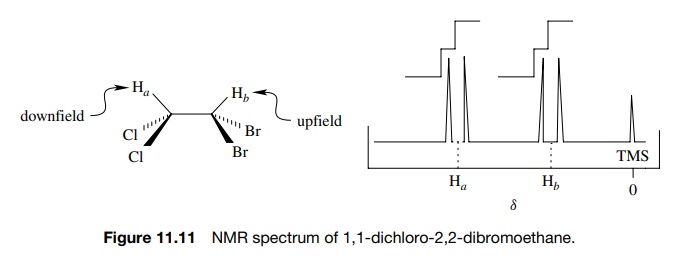
Consider
the NMR spectrum of 1,1-dichloro-2,2-dibromoethane (Figure 11.11). Based on the
different electronegativities of chlorine and bromine, the two protons in the
molecule are nonequivalent and should thus give signals at different chemical
shifts with the same integrated areas. The dichloromethyl proton should appear
downfield relative to the dibromomethyl proton. The actual NMR spectrum indeed
shows two different signals, one for Ha
and one for Hb , but each
absorption consists of two lines and is termed a doublet. The signal for each
proton is thus “split” into two resonances. This splitting is due to the fact
that each proton can sense the spin state of the neighboring proton and is called
spin – spin splitting.
In
the magnetic field of the NMR spectrometer, both Ha and Hb
have distinct absorption frequencies based on the Heff each proton experiences. This gives rise to
individual signals for Ha
and Hb . Focus now on one
hydrogen, Ha . The
hydrogen which is next to Ha
(namely Hb ) has two spin
states (aligned or opposed to Ho) that are nearly equally populated
(actually there are slightly more in the lower energy spin state than the
upper, but the difference is very small). Thus the magnetic moment of the
neighboring proton Hb (µHb ) either adds or subtracts an incremental amount (µHb ) to Heff
— the field experienced by Ha
. As a consequence Ha will
experience two distinct magnetic fields Heff
− µHb and Heff + µHb . Consequently Ha will absorb energy at two
distinct frequencies, and
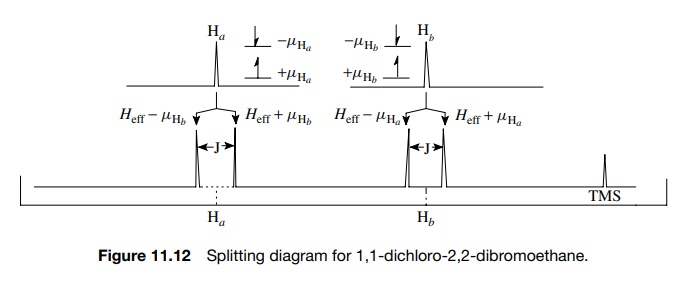
Focusing now on Hb , the same analysis leads
to the prediction that Hb
will also experience two distinct magnetic fields, Heff −
µHa and Heff
+ µHa ; absorb energy at two different frequencies; and thus will
be split into a doublet by the presence of Ha
(Figure 11.12).
The
middle of the doublet corresponds to the actual chemical shift of the proton
due to Heff, the total
integrated area under both lines of the doublet corresponds to the signal
intensity of one proton, and the width between the two lines in hertz (cps) is
called J , the coupling constant. The
coupling constant is a measure of the strength of the interaction between the
coupled nuclei that leads to spin – spin splitting. The J values for proton – proton coupling can range from 0 to 20 Hz,
but most commonly coupling constants fall in the range of 0 – 10 Hz.
A
value of J = 0 means that there is no
significant interaction with neigh-boring protons, and thus the absorption is
not affected by the spin states of neighboring protons. This normally occurs
when there are more than three bonds
separating different types of protons.
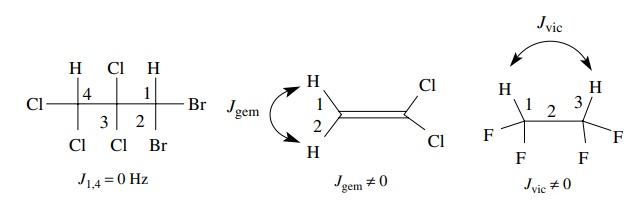
Geminal
coupling (two bonds, Jgem)
and vicinal coupling (three bonds, Jvic)
are the types of spin – spin splitting normally encountered. In addition, the
interaction between protons is reciprocal— if two protons are coupled, they are
coupled equally and J1,2 = J2,1. That is, if Ha is split by Hb by some amount, say J = 6 Hz, then Hb must be
split by Ha by J = 6 Hz. Finally equivalent protons do
not split each other; thus the t
-butyl hydrogens of t -butanol are a
singlet (Figure 11.13).
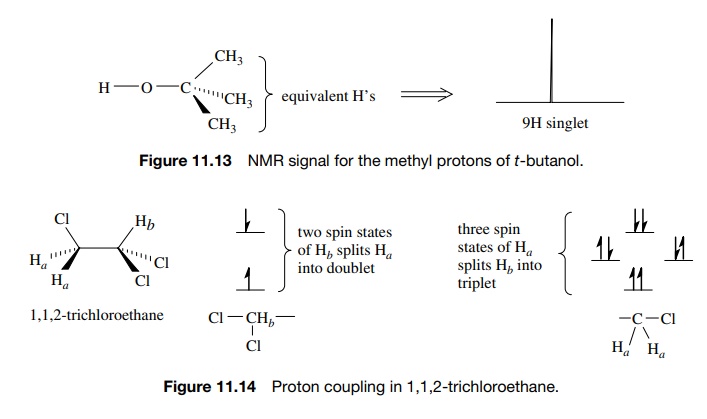
If
there are more than one neighboring hydrogen atom, then different split-ting
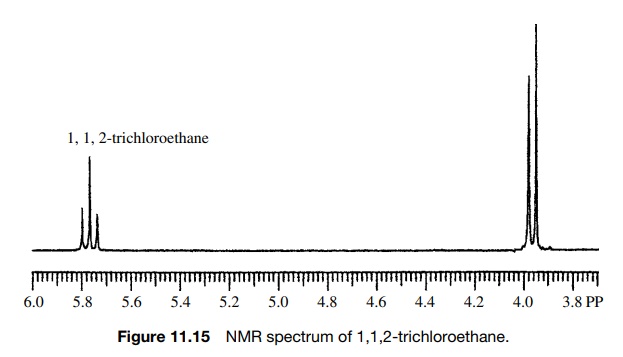
patterns are observed. For example, 1,1,2-trichloroethane has two signals, a
two-proton doublet (J = 6 Hz) upfield (Ha ) and a one-proton triplet
(J = 6 Hz) downfield (Hb ) (Figure 11.14). The equivalent
protons Ha of the CH2–Cl
group give the upfield absorption, which is split into a doublet by the single
vicinal proton Hb . The
single methine proton Hb
gives the downfield absorption, which is split into a triplet by the two
adjacent, equivalent methylene protons Ha
. The triplet splitting is due to the three spin distributions possible for the
two equiv-alent CH2 protons, both aligned, one aligned (two
possibilities), both opposed. The Hb
triplet has three lines in a 1 : 2 : 1 ratio, which reflects the numbers of
spin states of the neighboring CH2 group. Because the two sets of
protons are coupled, the spacing between each line of the triplet (J = 6 Hz) must be the same as the
doublet splitting (J = 6 Hz). The middle line of the
triplet corresponds to the chemical shift of the CH2 group, whereas
the middle of the doublet corresponds to the chemical shift of the methine
proton Hb (Figure 11.15).
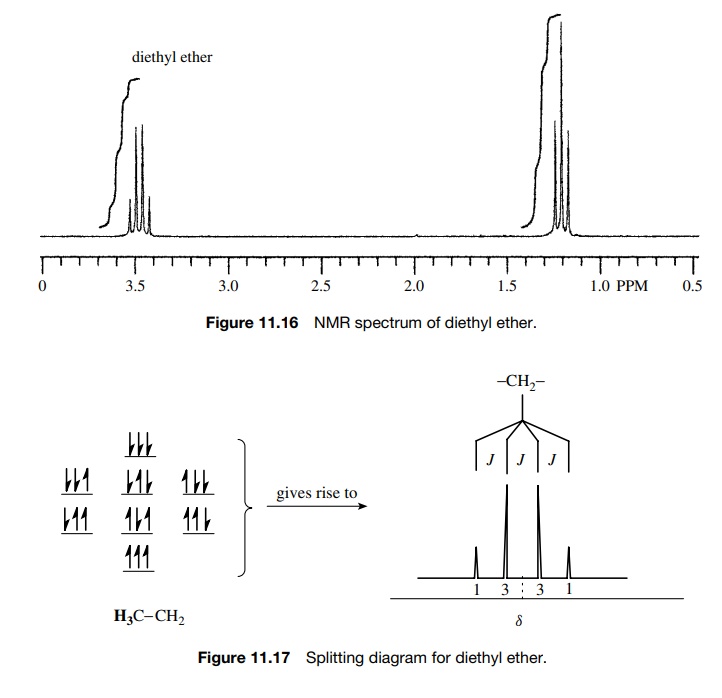
Diethyl
ether (Figure 11.16) has a three-proton triplet at 1.2δ (J = 7 Hz) for the methyl protons, which
are split by the two protons of the CH2 group. The methylene protons
absorb at 3.3δ and are split into
four lines (quartet) in a 1 : 3 : 3 : 1 ratio (Figure 11.17). This splitting
occurs because the three equiv-alent protons of the methyl group can have four
possible spin distributions which are nearly equally populated. They are three
aligned; two aligned, one opposed (three possibilities); one aligned, two
opposed (three possibilities); and all opposed. The center of the quartet is
the chemical shift of the CH2 group and the coupling constant of the
quartet (J = 7 Hz) must be the same as the
coupling constant of the methyl triplet (J
= 7 Hz) since the two
sets of protons are coupled. (When protons are coupled, the signal for each set
is split by the same coupling constant.)
If these considerations are generalized, it is seen that the signals for protons coupled equally to n equivalent vicinal protons will be split in to multiplets having n + 1 lines. The intensities of the individual lines in the multiplets follow
Pascal’s
triangle:

The
middle of the multiplet is the chemical shift of the protons responsible for
that absorption, and the total integrated area under the multiplet corresponds
to the total number of protons of the signal; however, the integrated area of
the individual lines of the multiplet are in the ratio of Pascal’s triangle.
Several examples of simple splitting patterns are shown:
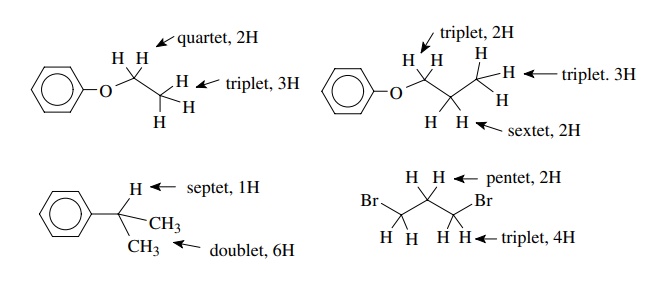
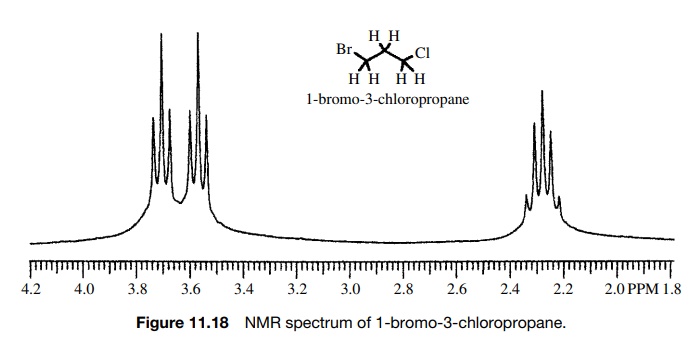
The
n + 1 rule for predicting the
multiplicity of a given proton signal holds when the coupling constants with
all of the nearest neighbors are the same. For example, the multiplicity of the
central methylene group of 1-bromo-3-chloropropane (Figure 11.18) is a pentet
which requires that J12 = J23.
That is, the central methylene group has the same coupling constant to the
protons of the bromomethyl group (J12)
as to the protons of the chloromethyl group (J23). Those groups are not equivalent and have different
chemical shifts, but each signal is split into a triplet by the C-2 methylene
group by the same J value.
If a proton or set of protons is not coupled equally to neighboring protons, then the n + 1 rule is not adequate to describe the multiplicity of the absorption. Instead one observes multiplets of multiplets as the splitting pattern (e.g., doublet of doublets or triplet of doublets). The multiplicity can be understood by carrying out sequential splitting diagrams. For example, consider a proton Hb split by two neighboring vicinal protons Ha and Hc by Jab = 2 Hz and Jbc = 7 Hz. This is shown schematically in Figure 11.19 where the Hb signal is split into a doublet by Hc (Jbc = 7 Hz) and each line of that doublet is split into a doublet by Ha (Jab = 2 Hz).
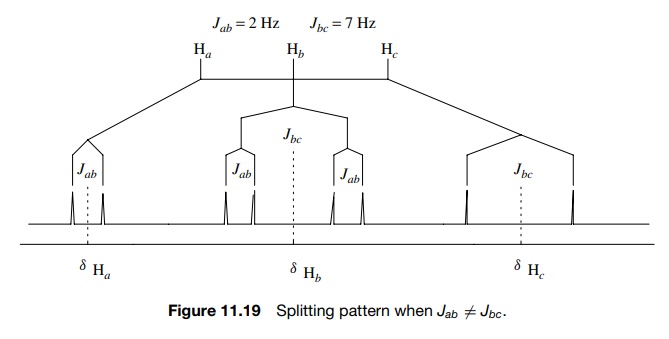
The result is a doublet of
doublets. The spacing between the small doublet splitting is J = 2 Hz and the splitting between the
centers of the two doublets is J = 7 Hz. The same diagram is produced
by first splitting the Hb
signal by Jab = 2 Hz and then splitting each line
into a doublet by Jbc = 7 Hz. Because of the requirement
that Jab = Jba
, Ha will be split into a
doublet (J = 2 Hz) by Hb and Hc
will also be split into a doublet (J = 7 Hz) by Hb . Taking into account these different splitting
patterns, the connectivity relationships between Ha , Hb
, and Hc are clear.
Because Ha and Hc are both doublets but they
are split by different coupling constants, they cannot be coupled to each
other. The signal for Hb ,
however, is seen to be a doublet of doublets with J = 2 Hz and J = 7 Hz. Since these
values are the same as the couplings of Ha
and Hc , Hb is coupled to both Ha and Hc and the connectivity is thus between Ha , Hb , and Hc
. Splitting patterns are thus powerful ways to establish connectivity in
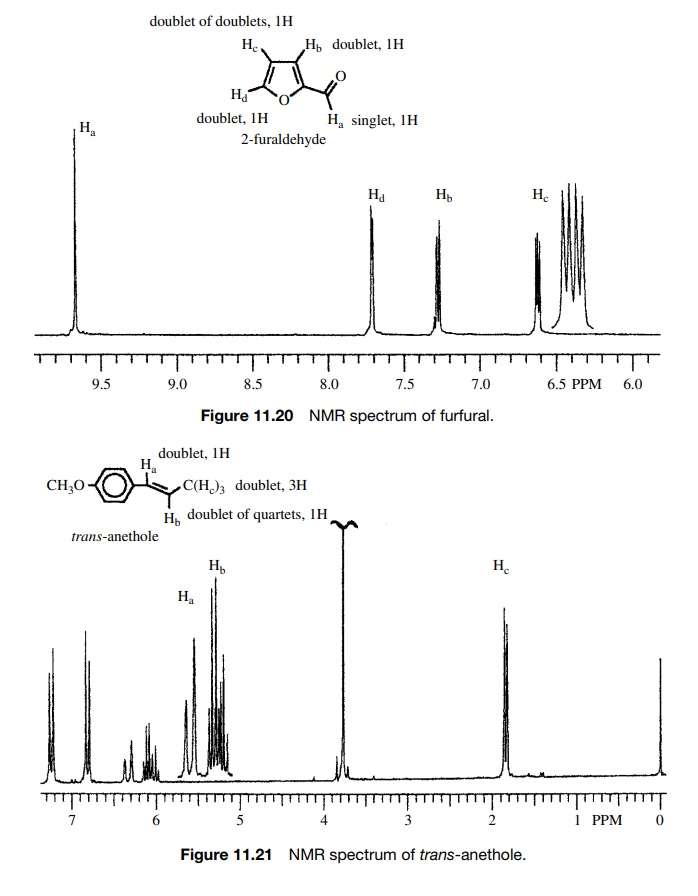
molecules. The patterns seen in Figures 11.20 and 11.21 are typical of the
types of connections encountered in various organic compounds.
Related Topics
