Structure of Skeletal Muscle Fibers
| Home | | Anatomy and Physiology | | Anatomy and Physiology Health Education (APHE) |Chapter: Anatomy and Physiology for Health Professionals: Support and Movement: Muscle Tissue
A single cell that can rapidly contract in response to stimulation and relaxes when the stimulation ceases is known as a skeletal muscle fiber.
Structure of
Skeletal Muscle Fibers
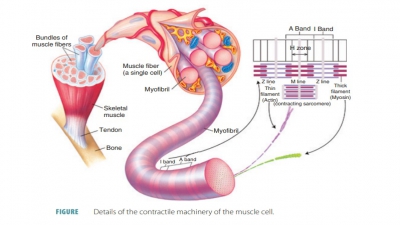
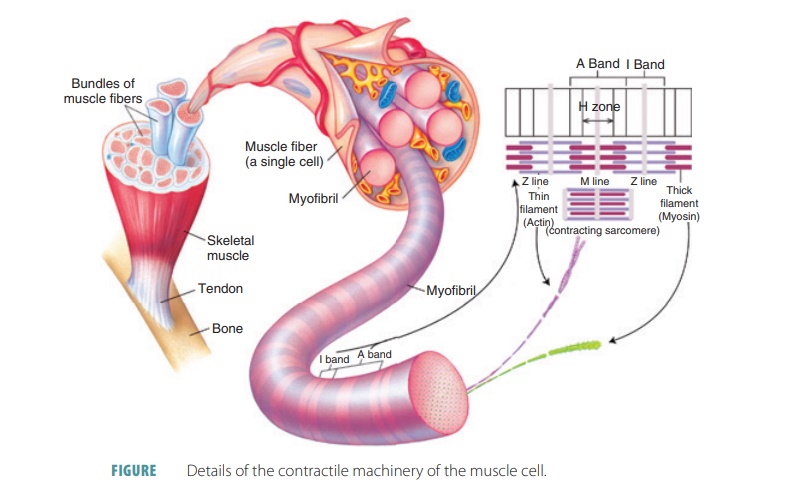
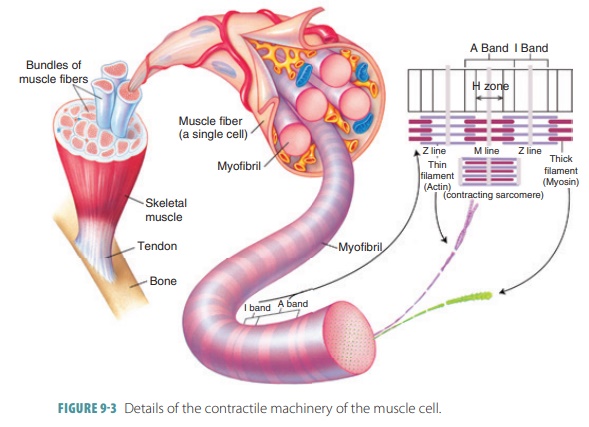
A single cell that can rapidly contract in response to stimulation and relaxes when the stimulation ceases is known as a skeletal muscle fiber (FIGURE 9-3). These fibers are thin, elongated cylinders with rounded ends. They are very large, with a diameter ranging from 10 to 100 μm. This is up to 10 times larger than an average body cell. The length may be up to 30 cm, and skeletal muscle fibers consist of hundreds of fused embryonic cells. The cell membrane (sarcolemma) lies above the cytoplasm (also known as sarcoplasm), with many contraction. The sarcoplasm also contains large amounts of glycosomes and myoglobin. Myoglobin is similar to hemoglobin, which is the pigment that transports oxygen in the blood. The collective struc-tures that comprise myofibrils make up approximately 80% of the cellular volume of muscle fibers.
Myofibrils
The sarcoplasm is made up of many
thread-like myofibrils arranged parallel to each other. One mus-cle fiber contains between
several hundred and several thousand rod-like myofibrils.
Filaments
Myofilaments have thick filaments composed of
myosin and thin filaments
composed of actin. These filaments are organized so they appear as striations— areas of alternating colored bands of skeletal muscle fiber. The
repeating patterns of striation units that appear along each muscle fiber are
referred to as sarcomeres, which are the functional units of skeletal muscle. Muscles are
basically considered to be collec-tions of sarcomeres.
Each skeletal muscle fiber
contains hundreds to thousands of myofibrils. Skeletal muscle fibers are the
longest types of muscle cells. They contain smaller, rod-shaped myofilaments.
The light bands (I bands) are made up of thin filaments of actinsmall, oval-shaped mitochondria and nuclei. Skeletal muscle
fibers have multiple nuclei. The advantage of these fibers is that they can
produce large amounts of the enzymes and structural proteins needed for
attached to round sheets known as Z lines (or Z
discs). The Z lines or discs are made
up of mostly alpha-actinin, a protein
that anchors the thin
filaments. The dark bands (A bands) are
made up of thick
filaments of myosin that overlap thin filaments of actin. The central
thick filaments contain myosin and extend the entire length of the A bands. The
thin filaments contain actin and extend across the I bands, part of the way
into the A bands. When a muscle fiber is intact, the A bands and I bands are
almost perfect in their alignment. The ability of a muscle to be stretched is
known as extensibility.
There is a central region (H zone) of thick fila-ments, with a
thickened area (the M line) that con-sists of proteins holding them in place. The H zone appears
less dense because the thin filaments do not extend into it. The M line is
slightly darker than the H zone because its fine protein strands hold the
nearby thick filaments together. The zone
of overlap is a dark area where thin filaments lie between thick filaments.
Three thick filaments surround every thin filament, and then six thin filaments
surround every thick filament. Myofilaments connect to the sar-colemma and are
held in an aligned pattern at the Z discs and M lines. Sarcomeres extend from
one Z line to another Z line. Other proteins form the structure of myofibrils. Elastic filaments are
com-posed of very large proteins known as titins. Each titin extends from the Z disc to the thick filament, forms the
core of the thick filament, and attaches to the M line. Titin binds thick
filaments in place, keeping the A bands organized and helps muscle cells to
return to normal shape after being stretched. This process is called elasticity. Dystrophin is an
important structural protein that links the thin fila-ments to the proteins of
the sarcolemma. Filaments and sarcomeres are also bound by proteins such as myomesin, nebulin, and C proteins.
Myosin molecules are made up of
two protein strands with globe-shaped cross-bridges that project outward.
Groups of many myosin molecules make up a myosin (thick) filament. Actin
molecules are globe-shaped with a binding site that attaches to myosin
cross-bridges. Groups of many actin molecules twist in double strands (helixes)
to form an actin (thin) fila-ment, which includes the proteins known as troponin and tropomyosin.
Polypeptide strands of the
rod-shaped tropo-myosin protein, at rest, prevent actin–myosin inter-action.
Tropomyosin spirals around the actin core, providing stiffening and
stabilization. One subunit of the round three-polypeptide complex troponin
protein molecule binds to tropomyosin, forming the troponin–tropomyosin
complex. Another subunit binds to G-actin to hold the complex in position. A third subunit has a receptor binding
a calcium ion. When the muscle is at rest, intracellular calcium is very low
and the binding site is empty. Contractions cannot occur unless the position of
the troponin– tropomyosin complex changes to expose the active sites on filamentous actin (F-actin). The
position change occurs when calcium ions bind to recep-tors on the troponin
molecules. When sarcomeres shorten within a skeletal muscle fiber, a skeletal
muscle contracts. This occurs because of the cross-bridges pulling on the thin
filaments of F- actin. Each strand of F-actin is made up of two rows of 300–400 globular molecules of G-actin.
Strands of tropomyo-sin cover the
G-actin active sites and prevent actin– myosin
interaction. A molecule of tropomyosin is a double-stranded protein covering
seven active sites, which is bound to one molecule of troponin halfway down its
length. A troponin molecule is made
up of three globular subunits.
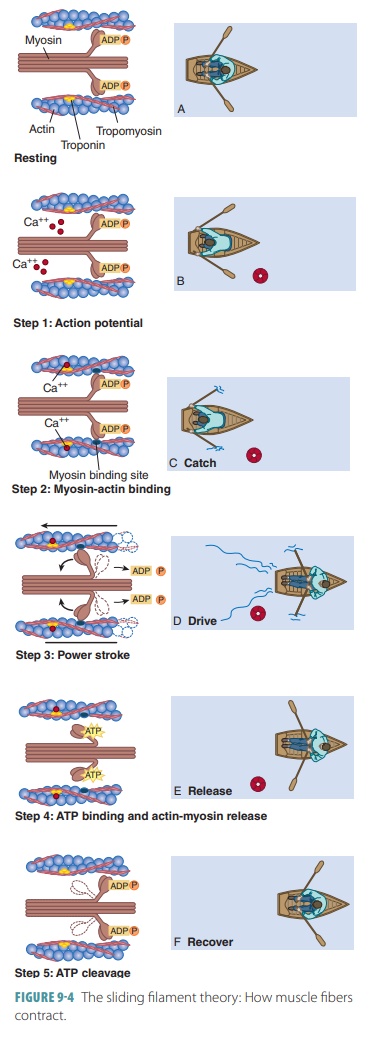
The sliding
filament modelor theory is so named because of
the way sarcomeres shorten, with thick and thin filaments sliding past each
other toward the center of the sarcomere, from both ends ( FIGURE 9 -4). If
cross-bridges generate enough tension on thin fil-aments, shortening occurs.
When the cross-bridges become inactive, contraction stops, tension decreases,
and the muscle fiber relaxes. Thin and thick filaments overlap only at the ends
of A bands. Therefore, the sliding filament model states that when contraction
occurs, thin filaments slide past thick filaments. Actin and myosin filaments
overlap more, and myosin on thick filaments connects with myosin-binding sites
on actin in the thin filaments. Cross-bridges are portions of myosin molecules.
Sliding begins, and cross-bridge attachments form and then break several times
during each contraction. They generate tension and move the thin filaments
toward the center of the sarcomere. Because this occurs throughout sarcomeres
in the cell at the same time, the muscle cell shortens. As thin filaments slide
centrally, attached Z discs are pulled toward the M line.
Myosin filaments contain the
enzyme ATPase in their globe-shaped portions. This enzyme catalyzes the breakdown of
ATP to both adenosine diphosphate (ADP) and phosphate, releasing energy. The
myosin cross-bridges act as ATPases during the contraction cycle of muscle.
They assume a “cocked” position, binding to actin to pull on the thin filament.
After the pulling occurs, the cross-bridge is released from actin before the
ATP splits. The cycle repeats as long as there is enough ATP for energy and
muscular stim-ulation occurs.
To understand further, the
following steps occur as a muscle cell shortens:
■■ I bands shorten.
■■ Distances between successive Z discs shorten.
■■ H zones (H bands) disappear.
■■ Contiguous A bands move closer together with out changing their length.
1. Describe
connective tissue associated with skeletal muscle tissue.
2. Describe
the structural components of a sarcomere.
3. Explain
the reason why skeletal muscle fibers appear striated.
Related Topics