Treatment of CHF
| Home | | Pharmacology |Chapter: Essential pharmacology : Cardiac Glycosides and Drugs for Heart Failure
Inotropic drugs—digoxin, dobutamine/ dopamine, amrinone/milrinone Diuretics—furosemide, thiazides
TREATMENT OF CHF
There are two distinct
goals of drug therapy in CHF:
a) Relief of congestive/low output symptoms
and restoration of cardiac performance:
Inotropic drugs—digoxin, dobutamine/ dopamine, amrinone/milrinone Diuretics—furosemide, thiazides
Vasodilators—ACE inhibitors/AT1 antagonists, hydralazine, nitrate,
nitroprusside
β
blocker—Metoprolol, bisoprolol, carvedilol
b) Arrest/reversal of disease progression and
prolongation of survival:
ACE inhibitors/AT1 antagonists
(ARBs)
β
blockers
Aldosterone antagonist—Spironolactone
Important
nonpharmacological measures are rest and salt restriction.
Rest reduces
peripheral needs, but should be advised only till compensation is restored,
beyond that it may lower myocardial reserve and be counterproductive. Salt
restriction limits edema formation and is advised in all grades of CHF. The
underlying cause of CHF, if treatable like hypertension, myocardial ischaemia,
valvular defects, AV shunts, arrhythmias, thyrotoxicosis, anaemia, should be corrected.
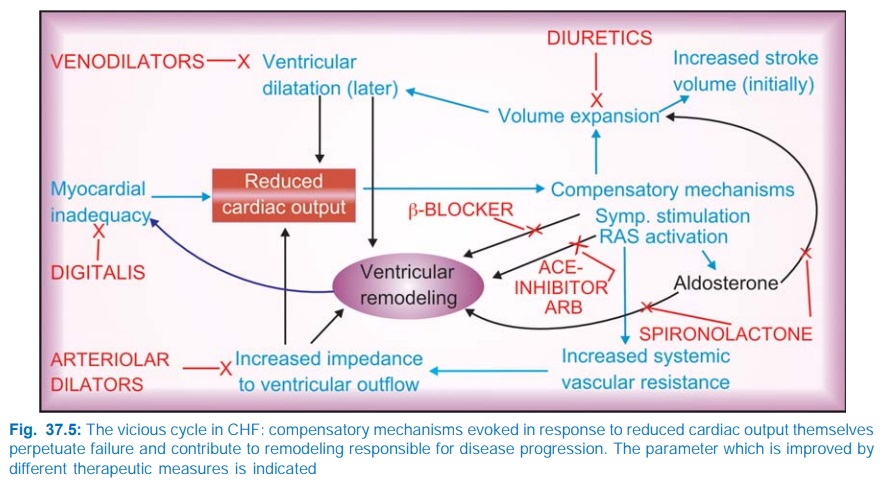
The pathophysiological
mechanisms that perpetuate heart failure and contribute to disease progression,
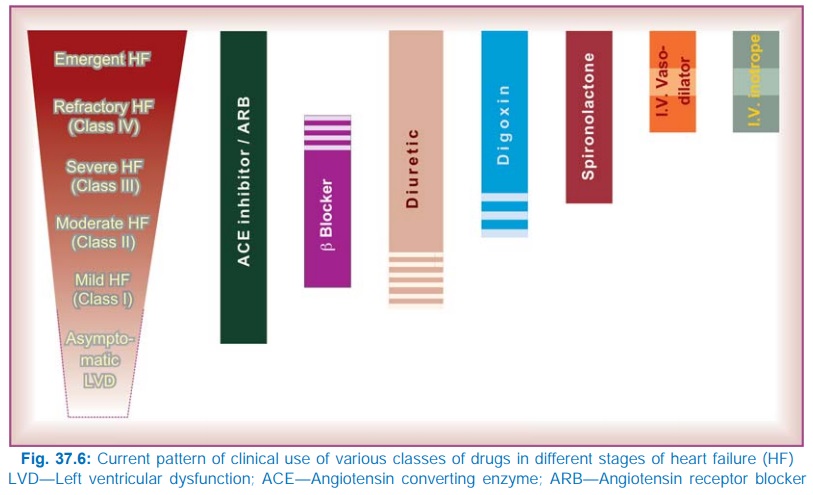
along with site of drug action are depicted in Fig. 37.5. The current pattern
of use of drugs in various stages of heart failure is summarized in Fig. 37.6.
Diuretics
Almost all cases of
symptomatic CHF are treated with a diuretic. High ceiling diuretics (furosemide, bumetanide) are the diuretics
of choice for mobilizing edema fluid; later they may be continued in low doses.
In advanced CHF after chronic use, resistance may develop to even high ceiling
diuretics: a thiazide/metolazone/spironolactone may be combined to overcome it.
Thiazide alone has
very limited role in CHF. Diuretics:
·
Decrease preload and improve ventricular efficiency
by reducing circulating volume.
·
Remove peripheral edema and pulmonary
congestion.
Intravenous furosemide
promptly increases systemic venous capacitance and produces rapid symptomatic
relief. It has, in conjunction with vasodilators, virtually obviated the need
for i.v. digitalization. Further, most mild cases can be maintained on
diuretics without recourse to chronic digitalis therapy. However, diuretics do
not influence the primary disease process in CHF, though they may dramatically
improve symptoms. Despite decades of experience, no prognostic benefit has been
demonstrated for diuretics. On the other hand, they may cause activation of
renin-angiotensin system (RAS) which has adverse cardiovascular consequences.
Chronic diuretic therapy tends to cause hypokalaemia, alkalosis and
carbohydrate intolerance. Current opinion is to treat mild heart failure with
ACE inhibitors/ARBs ± β blockers only, because they afford survival
benefit, while diuretics may be added intermittently as needed. Chronic
diuretic therapy should be reserved for relatively advanced cases with tendency
to fluid retention when diuretic is stopped. Dose should be titrated to the
lowest that will check fluid retention, but not cause volume depletion to
activate RAS.
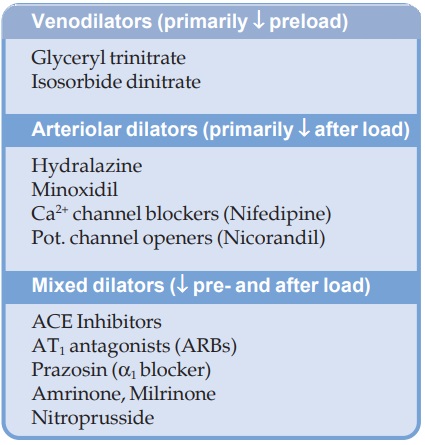
Vasodilators
Vasodilators are used i.v. to treat acute heart failure that
occurs in advanced cases, as well as orally for long-term therapy of chronic
CHF, and have become the mainstay of anti-CHF measures. Vasodilators with
differing profiles of arteriolar and venodilator action are available (see box).
1) Preload Reduction: Nitrates cause pooling of blood in systemic capacitance vessels and
reduce ventricular enddiastolic pressure and volume. With reduction in size of
ventricles, effectiveness of myocardial fibre shortening in causing ejection of
blood during systole improves (Laplace relationship). Controlled i.v. infusion
of glyceryl trinitrate affords rapid relief in acute left ventricular failure.
However, a marked lowering of preload (by vasodilators + strong diuretics) may
reduce output of a failing heart whose performance is dependent upon elevated
filling pressure. Occurrence of nitrate tolerance limits their utility in
routine treatment of CHF.
2) Afterload
Reduction: Hydralazine dilates resistance vessels and
reduces aortic impedance so that even weaker ventricular contraction is able to
pump more blood; systolic wall stress is reduced. It is effective in forward
failure when cardiac index (CI = min output/body surface area) is low (< 2.5
L/min/m2) without a marked increase in central venous pressure (< 18 mm Hg).
Marked tachycardia and fluid retention limit long-term use of
hydralazine monotherapy.
Trials of the three prototype calcium channel blockers
verapamil, diltiazem and nifedipine in systolic dysfunction have been
disappointing, even negative with occasional worsening of symptoms and increase
in mortality. This may be due to reflex sympathetic activation (nifedipine) or
negative inotropic property (verapamil, diltiazem).
Verapamil, however, is useful in diastolic dysfunction due to
hypertrophic cardiomyopathy. Trials with long-acting and more vasoselective
dihydropyridines (felodipine, amlodipine) have reported neither increase nor
decrease in heart failure mortality; may be used for symptomatic relief in
selected patients.
(iii) Pre and After Load Reduction:
ACE inhibitors/ ARBs
are orally active medium efficacy nonselective arteriovenous
dilators, while Sod. nitroprusside is high efficacy i.v.
dilator with equal action on the two
types of vessels. These drugs act by both the above mechanisms. Titrated i.v.
infusion of nitroprusside is employed in conjunction with a loop diuretic +
i.v. inotropic drug to tideover crisis in severely decompensated patients. For
symptomatic treatment of acute heart failure, choice of i.v. vasodilator
(glyceryl trinitrate or hydralazine or nitroprusside) depends on the primary
haemodynamic abnormality in individual patients.
In the long-term, survival benefit has been obtained only with a
combination of hydralazine + isosorbide dinitrate or with ACE inhibitors/ ARBs;
the latter performing better than the former. Only ACE inhibitors/ARBs alter the course of pathological changes in CHF
(see Ch. No. 36); afford symptomatic
as well as disease modifying benefits by retarding/reversing ventricular
hypertrophy, myocardial cell apoptosis and remodeling. Prognostic benefits of
ACE inhibitors/ARBs have been established in mild to severe (NYHA class I to IV)
CHF as well as in patients with asymptomatic systolic dysfunction. They are
thus recommended for all grades of CHF, unless contraindicated, or if renal
function deteriorates.
Hydralazine causes
more marked renal vasodilatation; may be selected for patients with renal
insufficiency who cannot tolerate ACE inhibitors. Severe CHF patients already
receiving ACE inhibitors + digoxin + diuretic have obtained extra benefit from
addition of hydralazine with or without a nitrate.
For reasons not known,
the α1 blocker prazosin has not been able to afford
prognostic benefit.
β-Adrenergic Blockers
Extensive studies over the past 25 years have now established
the utility of β1 blockers (mainly
metoprolol and bisoprolol) and the nonselective β + selective α1 blocker carvedilol in
mild to moderate CHF treated with ACE inhibitor ± diuretic/digitalis.
A large number of randomized trials including Metoprolol in
dilated cardiomyopathy trial (1993), US carvedilol trial (1996), MERITHF trial
(1999), CIBISII trial (1999), CAPRICORN trial (2001), COPERNICUS trial (2002)
have demonstrated subjective, objective, prognostic and mortality benefits of
the above 3 β blockers over and
above that afforded by ACE inhibitors + diuretic ± digitalis.
Though the immediate hemodynamic action of β blockers is to
depress cardiac contractility and ejection fraction, these parameters gradually
improve over weeks. After a couple of months ejection fraction is generally
higher than baseline, and slow upward titration of dose further improves
cardiac performance. The hemodynamic benefit is maintained over long-term and
hospitalization/mortality due to worsening cardiac failure, as well as all
cause mortality is reduced. The benefits appear to be due to antagonism of
ventricular wall stress enhancing, apoptosis promoting and pathological remodeling
effects of excess sympathetic activity in CHF, as well as due to prevention of
sinister arrhythmias. β blockers decrease plasma markers of
activation of sympathetic, renin-angiotensin systems and endothelin-1.
However, β blocker therapy in
CHF requires caution, proper patient selection and observance of several
guidelines:
·
Greatest utility of β blockers has been
shown in mild to moderate (NYHA class II, III) cases of dilated cardiomyopathy
with systolic dysfunction in which they are now routinely co-prescribed unless
contraindicated.
·
Encouraging results (upto 35% decrease in
mortality) have been obtained in class IV cases as well, but use in severe
failure could be risky and needs constant monitoring.
·
There is no place for β blockers in decompensated
patients. β blockers should be
stopped during an episode of acute heart failure and recommenced at lower doses
followed by up-titration after compensation is retored. Conventional therapy
should be continued along with them.
·
Starting dose should be very low—then titrated
upward as tolerated to target level (carvedilol 50 mg/day, bisoprolol 10 mg/day,
metoprolol 200 mg/day) or near it for maximum protection.
·
A long acting preparation (e.g. sustained
release metoprolol) or 2–3 times daily dosing to produce round the clock β blockade should be
selected.
·
There is no evidence of benefit in
asymptomatic left ventricular dysfunction.
Aldosterone Antagonist (Spironolactone)
Over the past 2 decades it has been realized that
rise in plasma aldosterone in CHF, in addition to its well known Na+ and water
retaining action, is an important contributor to disease progression by direct
and indirect effects:
a) Expansion of e.c.f.
volume → increased cardiac
preload.
b) Fibrotic change in myocardium → worsening systolic
dysfunction and pathological remodeling.
c) Hypokalemia and hypomagnesemia → increased risk of
ventricular arrhythmias and sudden cardiac death.
d) Enhancement of cardiotoxic effect of sympathetic
overactivity.
The aldosterone antagonist spironolactone is a weak diuretic (see Ch. No. 41), but can benefit CHF by
antagonizing the above effects of aldosterone.
In addition to several small studies, a large Randomised
aldactone evaluation study (RALES, 1999) conducted on 1663 NYHA class III and
IV patients having left ventricular ejection fraction < 35% has
confirmed the additional survival benefit (30%) of spironolactone when added to
conventional therapy with ACE inhibitors + other drugs. A subsequent trial
(EPHESUS, 2003) using another aldosterone antagonist eplerenone in post acute MI heart failure has further substantiated
the mortality and anti-remodeling benefit over and above that of ACE inhibitors
± β blockers.
Though ACE inhibitors themselves lower aldosterone levels, this
effect is incomplete and short lasting. Current evidence suggests the following
regarding spironolactone therapy in CHF:
· It is indicated as add-on therapy to ACE
inhibitors + other drugs in moderate-to-severe CHF.
· It can retard disease progression, reduce
episodes of decompensation and death due to heart failure as well as sudden
cardiac deaths, over and above the protection afforded by ACE inhibitors/ARBs ±
β blockers.
· Only low doses (12.5–25 mg/day) of spironolactone
should be used to avoid hyperkalaemia; particularly because of concurrent ACE
inhibitor/ARB therapy.
· It may help restoration of diuretic response
to furosemide when refractoriness has developed.
The onset of benefit
of spironolactone in CHF is slow. It is contraindicated in renal insufficiency:
carries risk of hyperkalemia—requires serum K+ monitoring. Gynaecomastia occurs
in a number of male patients. However, spironolactone is a significant
additional therapeutic measure in moderate-severe CHF with prognostic benefits.
Sympathomimetic Inotropic Drugs (see Ch. No. 9)
Drugs with β adrenergic and dopaminergic D1 agonistic
actions have positive inotropic and vasodilator properties through activation
of adenylyl cyclase which may be utilized to combat emergency pump failure.
Dobutamine (2–8 μg/kg/min) a relatively selective β1 agonist with
prominent inotropic action is the preferred drug for i.v. infusion in acute
heart failure accompanying myocardial infarction (MI), cardiac surgery as well
as to tide over crisis in advanced decompensated CHF.
Dopamine (3–10 μg/kg/min by i.v. infusion) has been used in cardiogenic shock due
to MI and other causes. While dobutamine does not raise (may lower) systemic
vascular resistance and is preferred in heart failure, dopamine tends to
increase afterload, especially at higher rates of infusion (>5 μg/kg/min) and has
limited utility in patients who are not in shock. Low rates of dopamine
infusion cause selective renal vasodilatation (D1 agonistic action)—improve
renal perfusion and g.f.r. This can restore diuretic response to i.v.
furosemide in refractory CHF. These drugs afford additional haemodynamic
support over and above vasodilators, digitalis and diuretics, but benefits are
shortlasting. Due to development of tolerance, these drugs have no role in the long-term
management of CHF.
Phosphodiesterase III Inhibitors
Theophylline is a phosphodiesterase inhibitor that is nonselective
for different isoforms of this enzyme which degrades intracellular cAMP and
cGMP. Intravenous aminophylline had been used in past for acute left
ventricular failure with limited benefits, but unacceptable toxicity.
Amrinone (Inamrinone)
It is chemically and pharmacologically
distinct from digitalis and catecholamines. This bipyridine derivative is a
selective phosphodiesterase III (PDE III) inhibitor. The PDE III isoenzyme is
specific for intracellular degradation of cAMP in heart, blood vessels and
bronchial smooth muscles. Amrinone increases myocardial cAMP and transmembrane
influx of Ca2+. It does not inhibit Na+K+ATPase, and its action is independent
of tissue catecholamines and adrenergic receptors.
The two most important actions of amrinone are positive inotropy and direct vasodilatation: has been called an
‘inodilator’. Compared to dobutamine, proportionately greater decrease in
systemic vascular resistance is noted.
In CHF patients i.v. amrinone action starts in 5 min and lasts
2–3 hours; elimination t½ is 2–5 hours. It increases cardiac index, left
ventricular ejection fraction and decreases peripheral vascular resistance,
CVP, left ventricular end diastolic volume and pressure accompanied by mild
tachycardia and slight fall in BP.
Adverse Effects
Thrombocytopenia is the most prominent and dose
related side effect, but is mostly transient and asymptomatic.
Nausea, diarrhoea, abdominal pain, liver damage, fever and
arrhythmias are the other adverse effects.
Use
Though amrinone is active orally, its oral use in maintenance therapy of CHF has been
abandoned, because efficacy was lost and mortality was increased in comparison
to placebo.
It is indicated only for shortterm i.v. use in severe and
refractory CHF, as an additional drug to conventional therapy with digitalis,
diuretics and vasodilators.
Dose: 0.5 mg/kg bolus
injection followed by 5–10 μg/kg/ min i.v. infusion (max. 10 mg/kg in 24
hours). AMICOR, CARDIOTONE 5 mg/ml (as lactate) 20 ml amp.
Milrinone Related to amrinone;
has similar action but is more
selective for PDE III, and is at least 10 times more potent. It is shorteracting
with a t½ of 40–80 min.
Thrombocytopenia is not significant. In long term prospective
trials, increased mortality has been reported with oral milrinone also.
Milrinone is preferred over amrinone for shortterm use.
Dose: 50 μg/kg i.v. bolus
followed by 0.4–1.0 μg/kg/min infusion.
PRIMACOR IV 10 mg/10
ml inj.
Nisiritide This recombinant brain
natriuretic peptide (BNP) has been
approved recently for i.v. use to relieve dyspnoea and other symptoms in
refractory CHF, especially in patients prone to develop cardiac arrhythmias. It
enhances salt and water excretion as well as produces vasodilatation.
Additional haemodynamic and symptomatic improvement can be obtained for shortperiods.
Related Topics
