Anthranilic acid derivatives (Fenamates)
| Home | | Medicinal Chemistry |Chapter: Medicinal Chemistry : Analgesics, Antipyretics, and NSAIDs
i. Flufenamic Acid (Arlef, Tarlef) ii. Mefenamic acid iii. Meclofenamate Sodium
Anthranilic
acid derivatives (Fenamates)
The
anthranilic acid class NSAIDs result from the application of classic medicinal
chemistry bioisosteric drug design concepts as these derivatives are nitrogen
isoteres of salicylic acid.
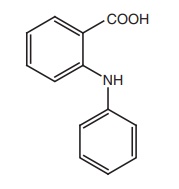
SAR of Anthranilic Acid Derivatives (Fenamates)
·The position of the carboxyl function is
important for the activity of anthranilic acid derivatives that are active,
whereas the 3 and 4 amino benzoic acid analogues are not active.
·Replacement of carboxylic acid function with the
isosteric tetrazole results in the retention of anti- inflammatory activity.
·Placement of substitution on the anthranilic
acid ring generally reduces the activity.
·Substitution on the N-aryl ring can lead to conflicting results. In the ultraviolet
erythema assay for anti-inflammatory activity, the order of activity was
generally 3´ > 2´ > 4´ for mono substitution with CF3 group
(flufenamic acid) being particularly potent. The opposite order of activity was
observed in rat paw oedema assay, the 2´–Cl derivatives being more potent than
3´–Cl analogues.
·In disubstituted derivatives, where the nature
of the two substitutes is the same 2´, 3´-disubstitution appears to be the most
effective (mefenemic acid).
·The NH moiety of anthranilic acid is essential
for the activity as the replacement of NH function with O, CH2, S, SO2, N-CH3,
or NCOCH3 functionalities significantly reduced the activity.
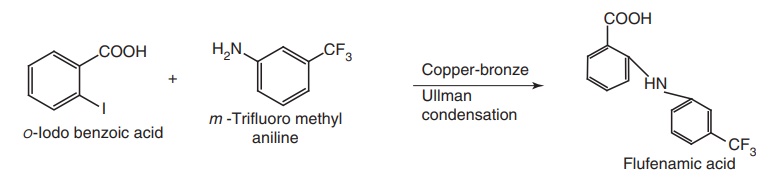
i. Flufenamic Acid (Arlef, Tarlef)
Synthesis
Properties and uses: Flufenamic acid is a pale yellow crystalline
powder or needles. It has analgesic, anti-inflammatory, and antipyretic actions;
it is employed in the treatment of rheumatic disorder and dysmenorrhoea.
Dose: 400–600 mg per day in divided doses.
ii. Mefenamic acid
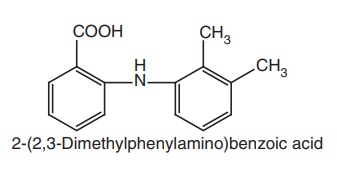
Synthesis
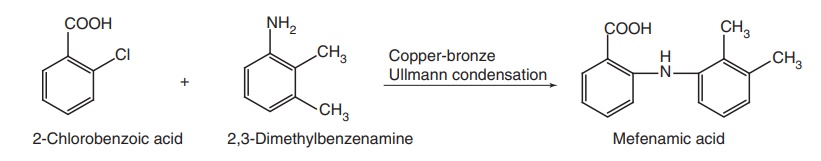
An analogues
approach by reaction of o-chloro
benzoic acid with 2,3-dimethyl aniline.
Metabolism: Its metabolism occurs through regioselective oxidation of
3-methyl group and glucuronidation of mephanamic acid. Majority of the
3-hydroxy methyl metabolite and dicarboxylic acid products are excreted.
Uses: Used as an analgesic and anti-inflammatory agent.
iii. Meclofenamate Sodium
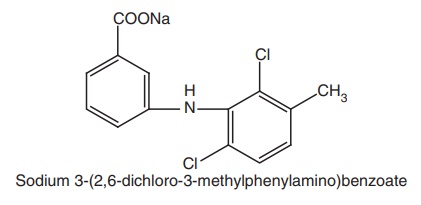
Synthesis: It is obtained by Ullman condensation employing 2, 6 dichloro
3-methyl aniline.
