Crystallization in Melts
| Home | | Pharmaceutical Technology |Chapter: Pharmaceutical Engineering: Crystallization
A melt may be defined as the liquid form of a single material or the homoge-neous liquid form of two or more materials that solidifies on cooling.
CRYSTALLIZATION IN MELTS
A
melt may be defined as the liquid form of a single material or the homoge-neous
liquid form of two or more materials that solidifies on cooling.
Crystal-lization in such a system is described by the following sequence: the
imposition of supercooling, the formation of nuclei, and the growth of
crystals.
If
a single-component liquid is cooled, some degree, often large, of supercooling
must be established before crystal nuclei are formed and growth begins. A
metastable liquid region exists below the melting point, which can only be
entered by cooling. In this metastable, supercooled region, the absence of
nucleation precludes the formation and growth of crystals. If, however, a
crystal seed is added, growth will occur. The deliberate seeding of a
metastable system is commonly employed in industrial crystallization. With
further cool-ing, spontaneous nucleation usually takes place and the released
heat of
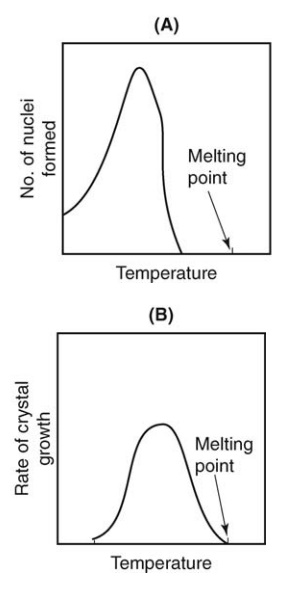
FIGURE 9.1 (A) Change in nucleation with
the degree of supercooling and (B) change in the rate of crystal growth with
the degree of supercooling.
crystallization
raises the temperature of the melt to its true melting point. With some
materials, lower temperatures increase the viscosity and prevent nuclea-tion.
The liquid then solidifies into a mass without crystallizing. This is known as
vitrification, and the products are called glasses. Many organic materials can
be obtained in this form, and, as with glass itself, devitrification may
suddenly occur, particularly after heating.
Related Topics
