Eicosanoids: Prostaglandins and Related Compounds
| Home | | Biochemistry |Chapter: Biochemistry : Phospholipid, Glycosphingolipid, and Eicosanoid Metabolism
Prostaglandins, and the related compounds thromboxanes and leukotrienes, are collectively known as eicosanoids to reflect their origin from polyunsaturated fatty acids with 20 carbons (eicosa = 20).
EICOSANOIDS: PROSTAGLANDINS AND RELATED COMPOUNDS
Prostaglandins, and the
related compounds thromboxanes and leukotrienes, are collectively known as
eicosanoids to reflect their origin from polyunsaturated fatty acids with 20
carbons (eicosa = 20). They are extremely potent compounds that elicit a wide
range of responses, both physiologic (inflammatory response) and pathologic
(hypersensitivity). They ensure gastric integrity and renal function, regulate
smooth muscle contraction (intestine and uterus are key sites) and blood vessel
diameter, and maintain platelet homeostasis. Although they have been compared
to hormones in terms of their actions, eicosanoids differ from endocrine
hormones in that they are produced in very small amounts in almost all tissues
rather than in specialized glands. They also act locally rather than after
transport in the blood to distant sites, as occurs with endocrine hormones such
as insulin. Eicosanoids are not stored, and they have an extremely short
half-life, being rapidly metabolized to inactive products. Their biologic
actions are mediated by plasma membrane G protein–coupled receptors, which are
different in different organ systems, and typically result in changes in cyclic
adenosine monophosphate production. Examples of prostaglandins and related
structures are shown in Figure 17.21.
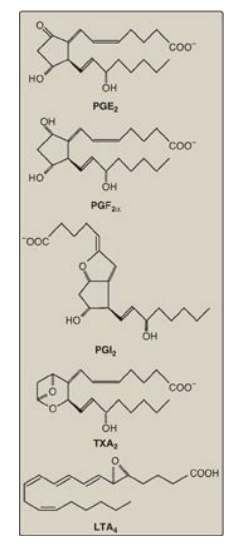
Figure 17.21 Examples of prostaglandin structures. [Note: Prostaglandins are named as follows: PG plus a third letter (for example, A, D, E, or F), which designates the type and arrangement of functional groups in the molecule. The subscript number indicates the number of double bonds in the molecule. PGI2 is known as prostacyclin. Thromboxanes are designated by TX and leukotrienes by LT.]
A. Synthesis of prostaglandins and thromboxanes
Arachidonic acid, an ω-6 fatty acid containing 20 carbons and four double bonds (an eicosatetraenoic fatty acid), is the immediate precursor of the predominant type of prostaglandins in humans (series 2 or those with two double bonds, as shown in Figure 17.22). It is derived by the elongation and desaturation of the essential fatty acid linoleic acid, also an ω-6 fatty acid. Arachidonic acid is incorporated into membrane phospholipids (typically PI) at carbon 2, from which it is released by phospholipase A2 in response to a variety of signals (Figure 17.23 ). [Note: Series 1 prostaglandins contain one double bond and are derived from an ω-6 eicosatrienoic fatty acid, dihomo-γ-linolenic acid, whereas series 3 contain three double bonds and are derived from eicosapentaenoic acid (EPA), an ω-3 fatty acid.]
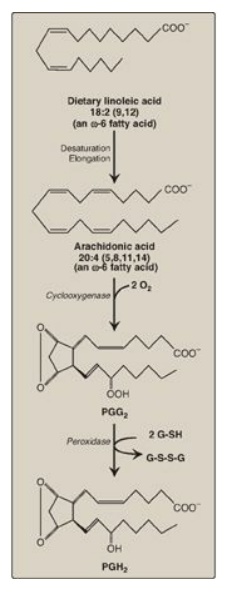
Figure 17.22 Oxidation and cyclization of arachidonic acid by the two catalytic activities (cyclooxygenase and peroxidase) of prostaglandin endoperoxide synthase. G-SH = reduced glutathione; G-S-S-G = oxidized glutathione.
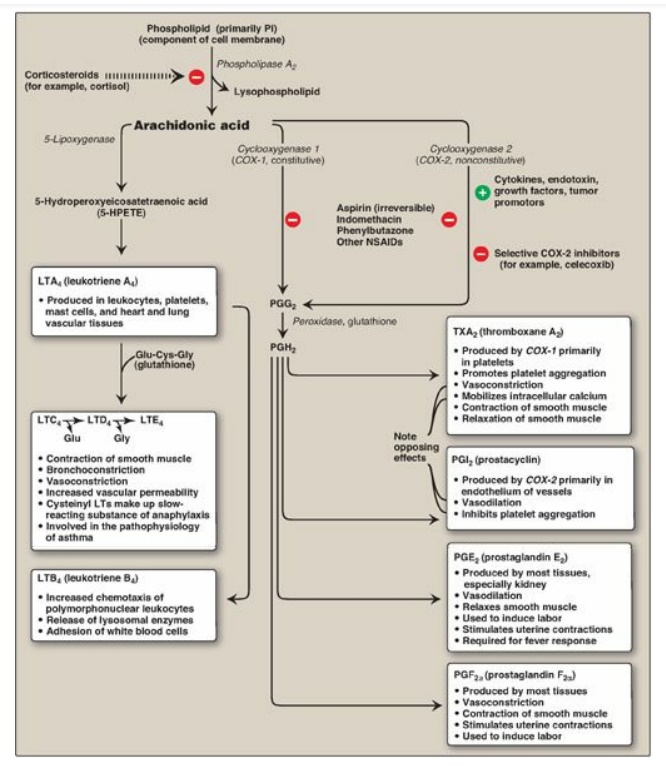
Figure 17.23 Overview of the biosynthesis and function of some important prostaglandins (PGs), leukotrienes (LTs), and a thromboxane (TX) from arachidonic acid. [Note: The arachidonic acid in the membrane phospholipid was derived from the ω-6 essential fatty acid, linoleic, also an ω-6 atty acid.] PI = phosphatidylinositol; NSAIDs = nonsteroidal anti-inflammatory drugs; Glu = glutamate; Cys = cysteine; Gly = glycine.
1. Synthesis of PGH2: The first step in prostaglandin
synthesis is the oxidative cyclization of free arachidonic acid to yield PGH2
by prostaglandin endoperoxide synthase (PGH synthase). This enzyme is an ER
membrane-bound protein that has two catalytic activities: fatty acid
cyclooxygenase (COX), which requires two molecules of O2, and
peroxidase, which is dependent on reduced glutathione. PGH2 is
converted to a variety of prostaglandins and thromboxanes, as shown in Figure
17.23, by cell-specific synthases.
a. Isozymes of PGH synthase: Two isozymes of PGH synthase,
usually denoted a s COX-1 and COX-2, are known. COX-1 is made constitutively in
most tissues and is required for maintenance of healthy gastric tissue, renal
homeostasis, and platelet aggregation. COX-2 is inducible in a limited number
of tissues in response to products of activated immune and inflammatory cells.
[Note: The increase in prostaglandin synthesis subsequent to the induction of
COX-2 mediates the pain, heat, redness, and swelling of inflammation and the
fever of infection.]
2. Inhibition of prostaglandin synthesis: The synthesis of prostaglandins can be inhibited by a number of unrelated compounds. For example, cortisol (a steroidal anti-inflammatory agent) inhibits phospholipase A2 activity (see Figure 17.23 ) and, therefore, the precursor of the prostaglandins, arachidonic acid, is not made available from membrane phospholipids. Aspirin, indomethacin, and phenylbutazone (all nonsteroidal anti-inflammatory drugs [NSAIDs]) inhibit both COX-1 and COX-2 and, thus, prevent the synthesis of the parent prostaglandin, PGH2. [Note: Systemic inhibition of COX-1, with subsequent damage to the stomach and the kidneys and impaired clotting of blood, is the basis of aspirin’s toxicity.] Aspirin (but not other NSAIDs) also induces synthesis of lipoxins and resolvins, lipid mediators with anti-inflammatory effects that are made from arachidonic acid and EPA, respectively. Inhibitors specific for COX-2 (the coxibs, for example, celecoxib) were designed to reduce pathologic inflammatory processes mediated by COX-2 while maintaining the physiologic functions of COX-1. However, their use has been associated with increased risk of heart attacks attacks, likely as a result of decreased PGI2 synthesis (see below), and some have been withdrawn from the market.
B. Synthesis of leukotrienes
Arachidonic acid is
converted to a variety of linear hydroperoxy (–OOH) acids by a separate pathway
involving a family of lipoxygenases (LOXs). For example, 5-lipoxygenase
converts arachidonic acid to 5-hydroperoxy-6,8,11,14 eicosatetraenoic acid
([5-HPETE], see Figure 17.23 ). 5-HPETE is converted to a series of
leukotrienes containing four double bonds, the nature of the final products
varying according to the tissue. Leukotrienes are mediators of allergic
response and inflammation. Their synthesis is not inhibited by NSAIDs. [Note:
Aspirin-induced asthma is a response to overproduction of leukotrienes with
NSAID use.] Inhibitors of 5-lipoxygenase and leukotriene receptor antagonists
are used in the treatment of asthma.
C. Role of prostaglandins in platelet homeostasis
Thromboxane A2 (TXA2)
is produced by COX-1 in activated platelets. It promotes adherence and
aggregation of circulating platelets and contraction of vascular smooth muscle,
thereby promoting formation of blood clots (thrombi). (See online Chapter 34.)
Prostacyclin (PGI2), produced by COX-2 in vascular endothelial
cells, inhibits platelet aggregation and stimulates vasodilation and, so,
impedes thrombogenesis. The opposing effects of TXA2 and PGI2
limit thrombi formation to sites of vascular injury. [Note: Aspirin has an
antithrombogenic effect. It inhibits TXA2 synthesis by COX-1 in
platelets and PGI2 synthesis by COX-2 in endothelial cells through irreversible
acetylation of these isozymes (Figure 17.24 ). The inhibition of COX-1 cannot
be overcome in platelets, which lack nuclei. However, the inhibition of COX-2
can be overcome in endothelial cells, because they have a nucleus and,
therefore, can generate more of the enzyme. This difference is the basis of
low-dose aspirin therapy used to lower the risk of stroke and heart attacks by
decreasing formation of thrombi.]
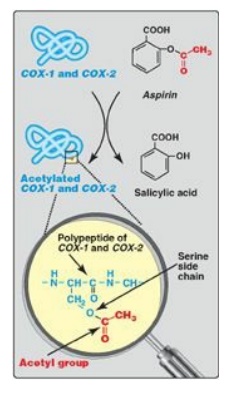
Figure 17.24 Irreversible acetylation of cyclooxygenase (COX)-1 and COX-2 by aspirin.
Related Topics
