Phospholipid Synthesis
| Home | | Biochemistry |Chapter: Biochemistry : Phospholipid, Glycosphingolipid, and Eicosanoid Metabolism
Glycerophospholipid synthesis involves either the donation of PA from cytidine diphosphate (CDP)-diacylglycerol to an alcohol or the donation of the phosphomonoester of the alcohol from CDP-alcohol to 1,2-diacylglycerol.
PHOSPHOLIPID SYNTHESIS
Glycerophospholipid
synthesis involves either the donation of PA from cytidine diphosphate
(CDP)-diacylglycerol to an alcohol or the donation of the phosphomonoester of
the alcohol from CDP-alcohol to 1,2-diacylglycerol (Figure 17.5 ). In both
cases, the CDP-bound structure is considered an “activated intermediate,” and
cytidine monophosphate (CMP) is released as a side product of
glycerophospholipid synthesis. A key concept in phosphoglyceride synthesis,
therefore, is activation, either of DAG or the alcohol to be added, by linkage
with CDP. [Note: This is similar in principle to the activation of sugars by
their attachment to uridine diphosphate (UDP).] The fatty acids esterified to
the glycerol alcohol groups can vary widely, contributing to the heterogeneity
of this group of compounds, with saturated fatty acids typically found at
carbon 1 and unsaturated ones at carbon 2. Most phospholipids are synthesized
in the smooth endoplasmic reticulum (ER). From there, they are transported to
the Golgi apparatus and then to membranes of organelles or the plasma membrane
or are secreted from the cell by exocytosis. [Note: Ether lipid synthesis from
dihydroxyacetone phosphate occurs in peroxisomes.]
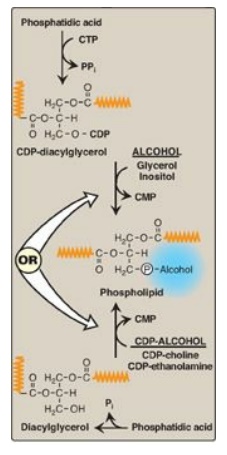
Figure 17.5 Phospholipid synthesis requires activation of either diacylglycerol or an alcohol by linkage to cytidine diphosphate (CDP). CMP = cytidine monophosphate; CTP = cytidine triphosphate; Pi = inorganic phosphate; PPi = pyrophosphate. (  is a fatty acid hydrocarbon chain.)
is a fatty acid hydrocarbon chain.)
A. Phosphatidic acid
PA is the precursor of
many other phosphoglycerides. The steps in its synthesis from glycerol
phosphate and two fatty acyl coenzyme A (CoA) molecules were illustrated in
Figure 16.14, in which PA is shown as a precursor of triacylglycerol.
Essentially all cells except mature erythrocytes
can synthesize phospholipids, whereas triacylglycerol synthesis occurs
essentially only in liver, adipose tissue, lactating mammary glands, and
intestinal mucosal cells.
B. Phosphatidylcholine and phosphatidylethanolamine
PC and PE are the most
abundant phospholipids in most eukaryotic cells. The primary route of their
synthesis uses choline and ethanolamine obtained either from the diet or from
the turnover of the body’s phospholipids. [Note: In the liver, PC also can be synthesized
from PS and PE (see below).]
1. Synthesis from preexisting choline and ethanolamine: These synthetic pathways involve the phosphorylation of choline or ethanolamine by kinases, followed by conversion to the activated form, CDP-choline or CDP-ethanolamine. Finally, choline-phosphate or ethanolamine-phosphate is transferred from the nucleotide (leaving CMP) to a molecule of DAG (see Figure 17.5).
a. Significance of choline reutilization: The reutilization of choline is
important because, whereas humans can synthesize choline de novo, the amount
made is insufficient for our needs. Thus, choline is an essential dietary
nutrient with an Adequate Intake of 550 mg for men and 425 mg for women. [Note:
Choline is also used for the synthesis of acetylcholine, a neurotransmitter.]
b. Role of phosphatidylcholine in lung surfactant: The pathway described above is the
principal pathway for the synthesis of dipalmitoylphosphatidylcholine (DPPC, or
dipalmitoyl lecithin). In DPPC, positions 1 and 2 on the glycerol are occupied
by palmitate. DPPC, made and secreted by type II pneumocytes, is a major lipid
component of lung surfactant, which is the extracellular fluid layer lining the
alveoli. Surfactant serves to decrease the surface tension of this fluid layer,
reducing the pressure needed to reinflate alveoli, thereby preventing alveolar
collapse (atelectasis). [Note: Surfactant is a complex mixture of lipids (90%)
and proteins (10%), with DPPC being the major component for reducing surface
tension.] Respiratory distress syndrome (RDS) in preterm infants is associated
with insufficient surfactant production and/or secretion and is a significant
cause of all neonatal deaths in Western countries.
Lung maturity of the fetus can be gauged by
determining the ratio of DPPC to sphingomyelin, usually written as the L (for
lecithin)-to-S ratio, in amniotic fluid. A value of two or above is evidence of
maturity, because it reflects the major shift from sphingomyelin to DPPC
synthesis that occurs in the pneumocytes at about 32 weeks of gestation.
Lung maturation can be
accelerated by giving the mother glucocorticoids shortly before delivery to
induce expression of specific genes. Postnatal administration of natural or
synthetic surfactant (by intratracheal instillation) is also used. [Note: Acute
respiratory distress syndrome (ARDS), seen in all age groups, is the result of
alveolar damage (due to infection, injury, or aspiration) that causes fluid to
accumulate in the alveoli, impeding the exchange of oxygen and carbon dioxide.]
2. Synthesis of phosphatidylcholine from
phosphatidylserine in the liver: The liver requires a mechanism for producing PC,
even when free choline levels are low, because it exports significant amounts
of PC in the bile and as a component of serum lipoproteins. To provide the
needed PC, PS is decarboxylated to PE by PS decarboxylase, an enzyme requiring pyridoxal
phosphate (PLP) as a coenzyme. PE then undergoes three methylation steps to
produce PC, as illustrated in Figure 17.6 . S-adenosylmethionine is the methyl
group donor.
C. Phosphatidylserine
PS synthesis in
mammalian tissues is provided by the base exchange reaction, in which the
ethanolamine of PE is exchanged for free serine (see Figure 17.6 ). This
reaction, although reversible, is used primarily to produce the PS required for
membrane synthesis.
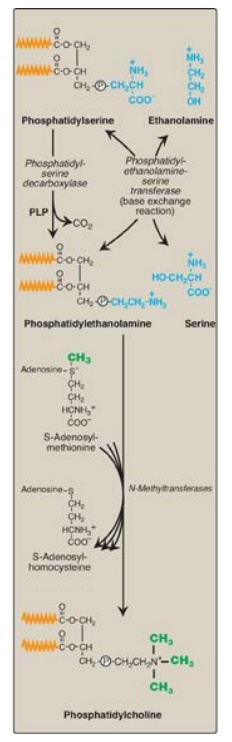
Figure 17.6 Synthesis of
phosphatidylcholine from phosphatidylserine in the liver. (  is a
fatty acid hydrocarbon chain.) P = phosphate; PLP
= pyridoxal phosphate.
is a
fatty acid hydrocarbon chain.) P = phosphate; PLP
= pyridoxal phosphate.
D. Phosphatidylinositol
PI is synthesized from free inositol and CDP-diacylglycerol as shown in Figure 17.5 . PI is an unusual phospholipid in that it most frequently contains stearic acid on carbon 1 and arachidonic acid on carbon 2 of the glycerol. PI, therefore, serves as a reservoir of arachidonic acid in membranes and, thus, provides the substrate for prostaglandin synthesis when required (see : for a discussion of these compounds). [Note: There is asymmetry in the phospholipid composition of the cell membrane. PS and PI, for example, are found primarily on the inner leaflet. Asymmetry is achieved by enzymes known as “flippases” and “floppases.”]
1. Role in signal transmission across membranes: The phosphorylation of
membrane-bound PI produces polyphosphoinositides such as phosphatidylinositol
4,5-bisphosphate ([PIP2] Figure 17.7 ). The hydrolytic cleavage of
PIP2 by phospholipase C occurs in response to the binding of a
variety of neurotransmitters, hormones, and growth factors to G protein–coupled
receptors (such as the α-1 adrenergic receptor) on the cell membrane and
activation of the Gq alpha subunit (Figure 17.8 ). The products of this
cleavage, inositol 1,4,5-trisphosphate (IP3) and DAG, mediate the mobilization
of intracellular calcium and the activation of protein kinase C, which act
synergistically to evoke specific cellular responses. Signal transmission across
the membrane is thus accomplished.
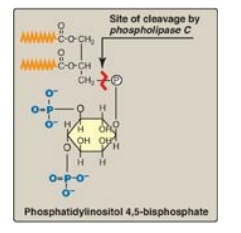
Figure 17.7 Structure of phosphatidylinositol 4,5-bisphosphate. Cleavage by phospholipase C produces inositol 1,4,5-trisphosphate and diacylglycerol. ( is a fatty acid hydrocarbon chain.) P = phosphate.
is a fatty acid hydrocarbon chain.) P = phosphate.
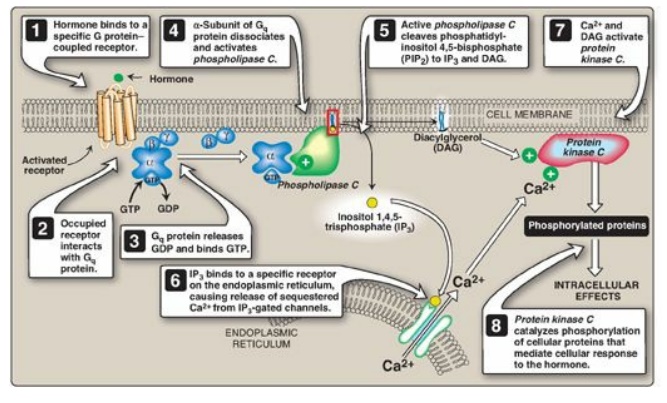
Figure 17.8 Role of inositol
trisphosphate and diacylglycerol in intracellular signaling. GTP = guanosine
triphosphate; GDP = guanosine diphosphate.
2. Role in membrane protein anchoring: Specific proteins can be covalently attached through a carbohydrate bridge to membrane-bound PI (Figure 17.9 ). Alkaline phosphatase, a digestive enzyme found on the surface of the small intestine that attacks organic phosphates, is an example of a protein attached to such a glycosyl phosphatidylinositol (GPI) anchor. [Note: GPI-linked proteins are also found in a variety of parasitic protozoans, such as, trypanosomes and leishmania.] Being attached to a membrane lipid (rather than being an integral part of the membrane) allows GPI-anchored proteins increased lateral mobility on the surface of the plasma membrane. The protein can be cleaved from its anchor by the action of phospholipase C (see Figure 17.8 ), releasing DAG. [Note: A deficiency in the synthesis of GPI in hematopoietic cells results in the hemolytic disease paroxysmal nocturnal hemoglobinuria.]
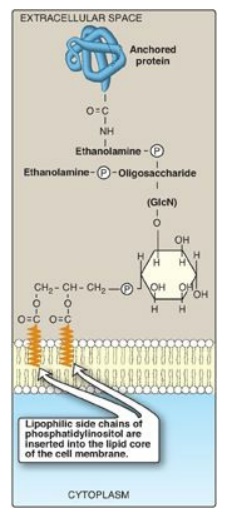
Figure 17.9 Example of a glycosyl phosphatidylinositol (GPI) membrane protein anchor. GlcN = glucosamine; P = phosphate.
E. Phosphatidylglycerol and cardiolipin
Phosphatidylglycerol occurs in relatively large amounts in mitochondrial membranes and is a precursor of cardiolipin (diphosphatidyglycerol). It is synthesized by a two-step reaction from CDP-diacylglycerol and glycerol 3-phosphate. Cardiolipin (see Figure 17.2) is synthesized by the transfer of diacylglycerophosphate from CDP-diacylglycerol to a preexisting molecule of phosphatidylglycerol.
F. Sphingomyelin
Sphingomyelin, a
sphingosine-based phospholipid, is a major structural lipid in the membranes of
nerve tissue. The synthesis of sphingomyelin is shown in Figure 17.10 .
Briefly, palmitoyl CoA condenses with serine, as CoA and the carboxyl group (as
CO 2) of serine are lost. [Note: This reaction, like the decarboxylation reactions
involved in the synthesis of PE from PS and of regulators from amino acids (for
example, the catecholamines from tyrosine) requires pyridoxal phosphate (a
derivative of vitamin B6) as a coenzyme.] The product is reduced in a
nicotinamide adenine dinucleotide phosphate (NADPH)-requiring reaction to
sphinganine (dihydrosphingosine), which is acylated at the amino group with one
of a variety of long-chain fatty acids and then desaturated to produce a
ceramide, the immediate precursor of sphingomyelin (and other sphingolipids).
Ceramides play a key role in maintaining the skin’s
water-permeability barrier.
Decreased ceramide levels are associated with a
number of skin diseases.
Phosphorylcholine from
PC is transferred to the ceramide, producing sphingomyelin and DAG. [Note:
Sphingomyelin of the myelin sheath contains predominantly longer-chain fatty
acids such as lignoceric acid and nervonic acid, whereas gray matter of the
brain has sphingomyelin that contains primarily stearic acid.]
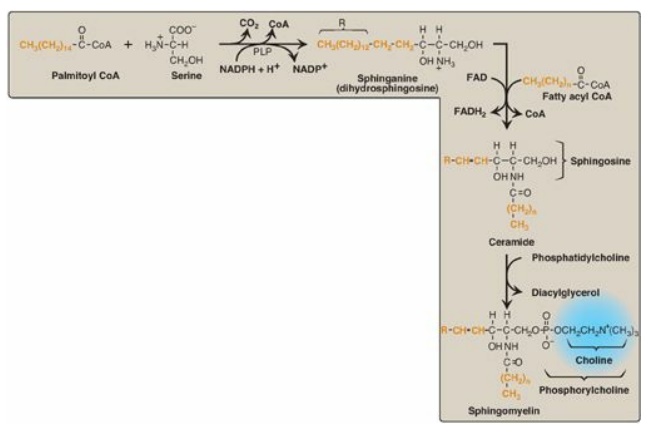
Figure 17.10 Synthesis of
sphingomyelin. PLP = pyridoxal phosphate; NADP(H) = nicotinamide adenine
dinucleotide phosphate; FAD(H2) = flavin adenine dinucleotide; CoA =
coenzyme A.
Related Topics
