Filtration Sterilization - Sterilization Methods
| Home | | Pharmaceutical Microbiology | | Pharmaceutical Microbiology |Chapter: Pharmaceutical Microbiology : Sterilization Procedures And Sterility Assurance
The process of filtration is unique among sterilization techniques in that it removes, rather than destroys, microorganisms. Further, it is capable of preventing the passage of both viable and nonviable particles and can thus be used for both the clarification and sterilization of liquids and gases.
FILTRATION STERILIZATION
The process
of filtration is unique among
sterilization techniques in that it removes,
rather than destroys, microorganisms. Further, it is capable of
preventing the passage of both viable
and nonviable particles and can thus be used for
both the clarification and sterilization of liquids and gases.
The principal application of sterilizing grade
filters is the treatment of heat-sensitive injections and ophthalmic solutions, biological products and air
and other gases for supply to aseptic areas.
Filters may also be required in industrial applications where they become
part of venting
systems on fermenters, centrifuges, autoclaves and
freeze-driers. Certain types of filter (membrane filters) also have an important
role in sterility testing, where they can be employed
to trap and concentrate contaminating organisms from solutions under test. These
filters are then placed on a
solid nutrient medium
or in a liquid medium and incubated to encourage colony growth or turbidity.
The major mechanisms of
filtration are sieving, adsorption and trapping
within the matrix
of the filter material. Of these,
only sieving can be regarded as absolute as it ensures
the exclusion of all particles above a defined size. It is generally accepted that synthetic
membrane filters,
derived from cellulose esters or other
polymeric materials, approximate most closely
to sieve filters; while fibrous pads,
sintered glass and sintered ceramic products can be regarded
as depth filters
relying principally on
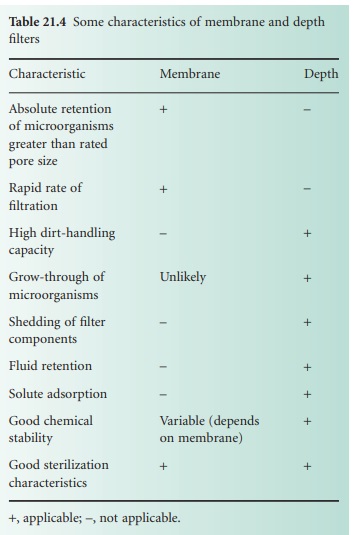
mechanisms of adsorption and entrapment. Some
of the characteristics of filter
media are summarized in Table 21.4.
The potential hazard
of microbial multiplication within a depth filter and subsequent
contamination of the filtrate
(microbial grow-through) should be recognized.
a) Filtration Sterilization Of Liquids
In order to compare
favourably with other methods of sterilization, the microorganism
removal efficiency of filters employed in the processing of liquids must be high. For
this reason, membrane
filters of 0.2–0.22
μm nominal pore diameter are
chiefly used, while
sintered filters are used only in restricted circumstances, i.e. for the
processing of corrosive
liquids, viscous fluids
or organic solvents. It may be tempting to assume that the pore
size is the major determinant of filtration efficiency and two filters of 0.2 μm pore diameter from different manufacturers will behave similarly. This
is not so, because, in addition to the sieving effect,
trapping within the
filter matrix, adsorption and charge effects all contribute
significantly towards the removal
of particles. Consequently, the depth of the membrane, its charge and the tortuosity of the channels are all factors which can make the performance of one filter far superior
to that of another.
The major criterion by which filters should be compared,
therefore, is their titre reduction values, i.e. the ratio of the number of organisms challenging a filter under
defined conditions to the number penetrating it. In all cases, the filter medium employed must be sterilizable, ideally by steam
treatment; in the case of membrane filters
this may be for once-only use, or, in the case of larger industrial filters, a small, fixed number of resterilizations; sintered
filters may be resterilized many times. Filtration
sterilization is an aseptic process
and careful monitoring of filter integrity is necessary as well as final
product sterility testing.
Membrane filters, in the form
of discs, can
be assembled into pressure-operated
filter holders for syringe mounting and in-line
use or vacuum
filtration tower devices. Filtration under pressure is generally considered most suitable, as filling at high flow
rates directly into
the final containers is possible without
problems of foaming, solvent evaporation or air leaks. To increase
the filtration area, and hence process
volumes, several filter
discs can be used in parallel
in multiple-plate filtration systems or, alternatively, membrane filters can be
fabricated into plain or pleated cylinders and installed in cartridges.
Membrane filters are often used in combination with a coarse-grade fibre glass depth prefilter to improve their dirt-handling
capacity.
b) Filtration Sterilization Of Gases
The principal
application for filtration sterilization of gases is in the provision of sterile air to aseptic
manufacturing suites,
hospital isolation units and some operating
theatres. Filters
employed generally consist
of pleated sheets of glass
microfibres separated and supported by an
aluminium framework; these
are employed in ducts, wall or ceiling panels,
overhead canopies, or laminar airflow cabinets. These high-efficiency particulate air (HEPA)
filters can remove up to 99.997%
of particles more than 0.3 μm in diameter and thus are
acting as depth filters. In practice, their microorganism removal efficiency is rather better
as the majority of bacteria are found associated with
dust particles and
only the larger fungal spores are found in the free state. Air is forced through HEPA filters by blower fans, and prefilters are used to remove
larger particles to extend the lifetime of the
HEPA filter. The operational efficiency and integrity of a HEPA filter can be monitored by pressure differential and airflow rate measurements,
and dioctylphthalate smoke particle
penetration tests.
Other applications of
filters include sterilization of venting or displacement air in tissue and microbiological culture (carbon filters and hydrophobic membrane filters); decontamination of air in mechanical
ventilators (glass fibre filters); treatment
of exhausted air from
microbiological safety
cabinets (HEPA filters); and the clarification
and sterilization of medical gases (glass wool depth filters and hydrophobic membrane filters).
Related Topics
