Interactions Affecting Absorption, Distribution, Metabolism, Excretion of Drugs
| Home | | Biopharmaceutics and Pharmacokinetics |Chapter: Biopharmaceutics and Pharmacokinetics : Pharmacokinetic Drug Interactions
Interactions Affecting Absorption of Drugs, Interactions Affecting Distribution of Drugs, Interactions Affecting Metabolism of Drugs, Interactions Affecting Excretion of Drugs
Interactions Affecting Absorption of Drugs
Altered absorption after oral administration is
very common. The interaction may result in a change in the rate of absorption
(an increase or a decrease), a change in the amount of drug absorbed (an
increase or a decrease) or both. Several mechanisms may be involved in the
alteration of drug absorption from the GIT. In general, drugs that are not
absorbed completely/rapidly are more susceptible to changes in GI absorption. A
decrease in the rate of absorption is clinically significant in acute
conditions such as pain where the drug is administered in a single dose but is
of little importance for drugs used in chronic therapy.
An alteration in parenteral drug absorption is rare
but can occur when an adrenergic agent such as adrenaline or a cholinergic drug
such as methacholine is extravascularly injected concomitantly with another
drug. These agents alter the systemic absorption of the latter due to
vasoconstriction or vasodilation.
Interactions Affecting Distribution of Drugs
Though several factors govern the distribution of
drugs to various tissues, clinically significant interactions result due to
competition between drugs for binding to proteins/tissues and displacement of
one drug by the other. Competitive displacement, which results when two drugs
are capable of binding to the same site on the protein, causes the most
significant interactions. Greater risk of interactions exists when the
displaced drug is highly protein bound (more than 95%), has a small volume of
distribution and has a narrow therapeutic index (e.g. tolbutamide, warfarin and
phenytoin), and when the displacer drug has a higher degree of affinity than
the drug to be displaced. In such situations, displacement of even a small
percent of drug results in a tremendous increase in the free form of the drug,
which precipitates increased therapeutic or toxic effects.
Drugs may also be displaced from binding sites in
tissues. An interesting example of this is oral hypoglycaemics such as the
sulphonyl ureas (tolbutamide, glibenclamide, etc.). These agents exert their
therapeutic effects by displacing insulin from protein binding sites in
pancreas, plasma and other regions resulting in its elevated levels.
Interactions Affecting Metabolism of Drugs
The most important and the most common cause of
pharmacokinetic interactions is alteration in the rate of biotransformation of
drugs. Major problems arise when one drug either induces or inhibits the
metabolism of another drug. Even the environmental chemicals can bring about
such an effect. The influence of enzyme inducers and inhibitors become more
pronounced when drugs susceptible to first-pass hepatic metabolism are given
concurrently. The metabolic pathway usually affected is phase I oxidation.
Enzyme inducers reduce the blood level and clinical efficacy of co-administered
drugs but may also enhance the toxicity of drugs having active metabolites. In
contrast to enzyme induction, which is usually not hazardous, enzyme inhibition
leads to accumulation of drug to toxic levels and serious adverse effects may
be precipitated.
Interactions Affecting Excretion of Drugs
Clinically significant renal excretion interactions
occur when an appreciable amount of drug or its active metabolite(s) are
eliminated in the urine. Excretion pattern can be affected by alteration in
GFR, renal blood flow, passive tubular reabsorption, active tubular secretion
and urine pH. An interesting pharmacokinetic interaction that results due to
the pharmacodynamic drug effect is between thiazide diuretics and lithium.
Owing to the influence of former on the renal tubular transport of sodium, the
lithium ions are retained in the body resulting in its toxicity.
Biliary excretion, the other major mechanism of
drug excretion, is altered by agents that inhibit biliary transport or modify
bile flow rate.
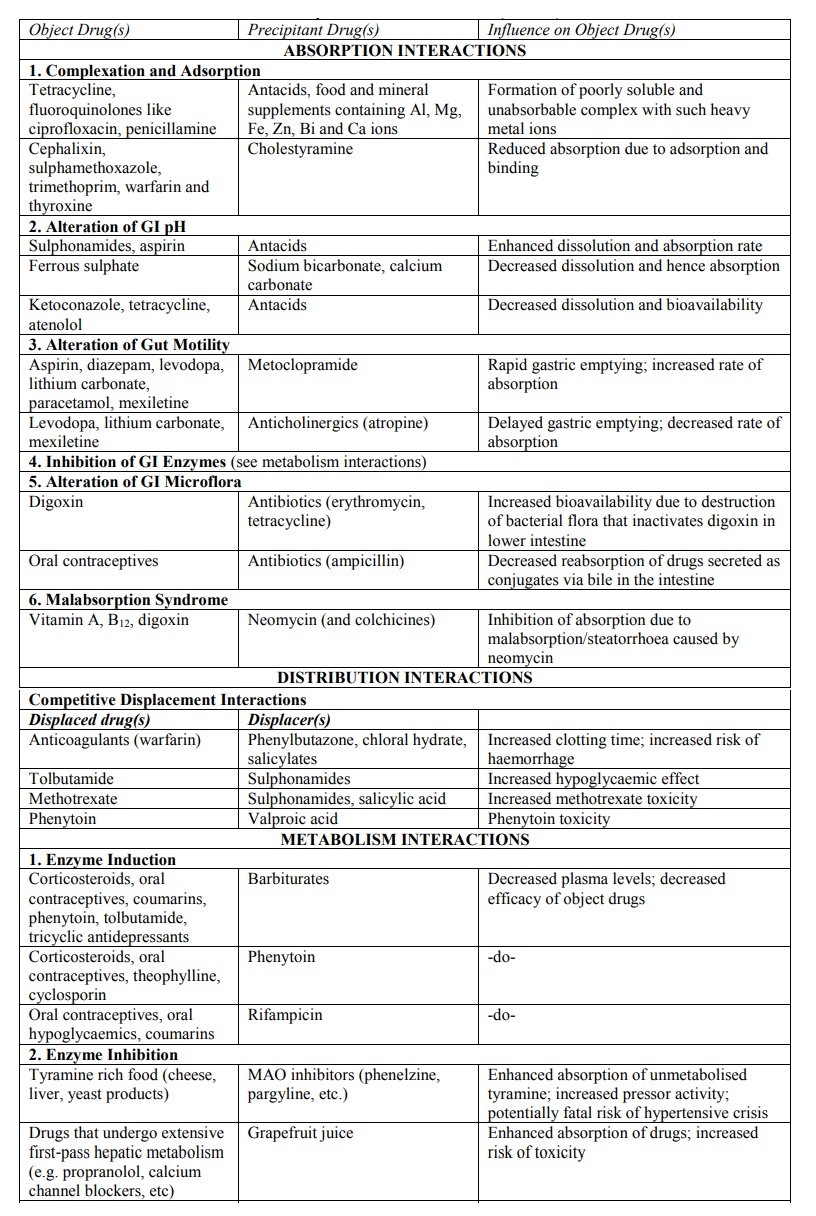
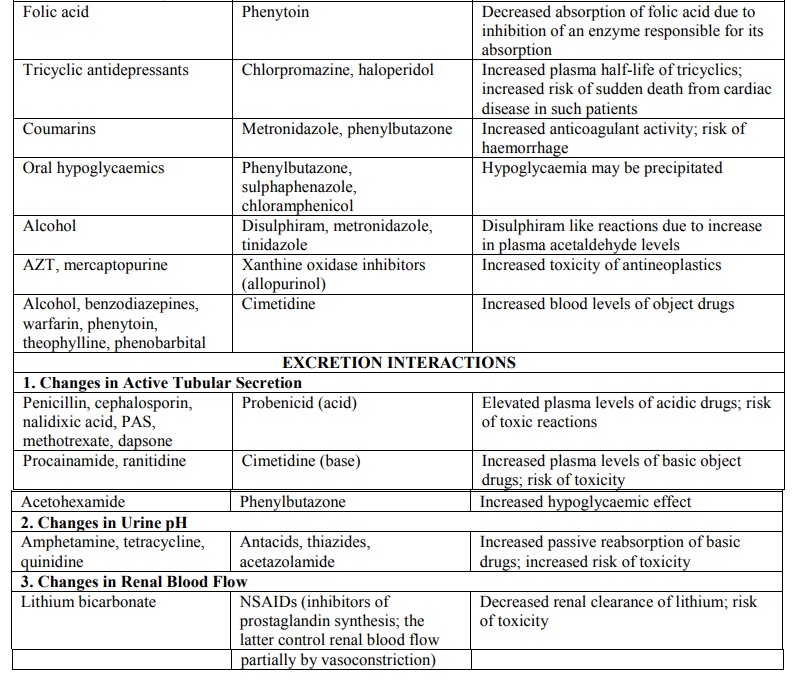
TABLE 7.1
List of Some of the Important Pharmacokinetic (ADME) Interactions
Reducing the Risk of Drug Interactions – Principles of Drug Interactions
Management The consequences of drug interactions may be –
·
Major – life threatening
·
Moderate – deterioration of
patient’s status
·
Minor – bothersome or little
effect.
The risk of drug interactions is a challenge that
embraces a number of considerations.
The following are guidelines to reduce and manage
drug interactions.
1. Identify
patient risk factors such as age, the nature of the patient's medical problem
(e.g., impaired renal function), dietary habits, smoking, and problems such as
alcoholism influence the effect of certain drugs.
2. Take
thorough drug history and maintain complete patient medication records.
3. Keep
knowledge about actions (both primary and secondary pharmacological actions) of
drugs being utilized.
4. Consider
therapeutic alternatives.
5. Avoid
complex therapeutic regimens where possible.
6. Educate
the patient to comply with instructions for administering medications. They
should be encouraged to ask questions about their therapy and to report any
excessive or unexpected responses.
7. Monitor
therapy: Any change in patient behavior should be suspected as drug-related
until that possibility is excluded.
8. Individualize
therapy: priority should be assigned to the needs and clinical response of the
individual patient, rather than to the usual dosage recommendations, standard
treatment, and monitoring guidelines.
9. Involve
the patient as a partner in health care. If the optimal benefits of therapy are
to be achieved with minimal risk, each participant must be knowledgeable about
and diligent in fulfilling his responsibilities.
Related Topics
