Resonance
| Home | | Organic Chemistry |Chapter: Organic Chemistry : Functional Groups and Chemical Bonding
Valence shell electrons of the atoms in a molecule are either shared or unshared. The shared electrons are found in either σ or π bonds. Unshared electrons are found in AOs (usually hybrid AOs for first-row elements).
RESONANCE
Valence
shell electrons of the atoms in a molecule are either shared or unshared. The
shared electrons are found in either σ
or π bonds. Unshared electrons are
found in AOs (usually hybrid AOs for first-row elements). Lewis struc-tures
provide a way to indicate the shared and unshared pairs of electrons in
molecules. Sometimes, however, it is possible to indicate the electron
distri-bution in molecules by more than one Lewis structure. For example, a
car-boxylate anion can be represented by two equivalent but different Lewis
structures.
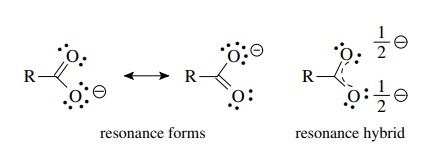
These
structures are equivalent because they have the same numbers of bonds, unshared
pairs of electrons, and the same charge. They are different because the
negative charge is located on different oxygen atoms. Moreover the bonds from
carbon to a particular oxygen are double in one structure and single in the
other. When more than one correct Lewis structure can be written for a
molecule, each structure is a resonance form of the molecule. The actual
molecule is a resonance hybrid of the contributing resonance forms, and its properties
result from a combination of the properties of the contributing resonance
forms. Thus each oxygen atom carries a – 1/2 charge, and the bonds between
carbon and each oxygen atom have a bond order of 1.5 and are of the same
length.
A
very good analogy is a mule. A mule is a hybrid of a horse and a donkey. A mule
is neither a horse nor a donkey but it has properties of each. The reso-nance
hybrid of the carboxylate anion is a resonance hybrid of the contributing
resonance forms and has properties of each.
Another
classic example of resonance is the benzene molecule. The localized resonance
forms are termed Kekule´ forms (after Friedrich August Kekule,´ who first
deduced the structure of benzene) and have alternating single and double bonds
between carbon atoms. The actual benzene molecule is a resonance hybrid of the
contributing resonance forms as the bond lengths are equal (single and double
bonds have different lengths).
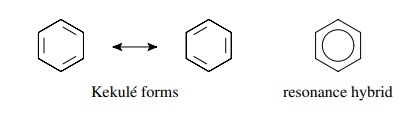
The
bond order is between one (single) and two (double). The resonance hybrid is
often pictured with a circle in the ring to indicate the delocalized electron
distribution in the molecule.
Double-headed
arrows are used to indicate resonance forms. It is important to note that
resonance forms are not in equilibrium, just as a mule is not a horse part of
the time and a donkey the rest of the time.
The
presence of resonance forms means that the electrons are not localized between
two nuclei but are delocalized over more than two nuclei. The result of electron
delocalization is that electrons are attracted by a greater number of nuclei,
which leads to a lower energy for the molecule and hence greater stability.
Simply put, resonance delocalization is a stabilizing feature of molecules.
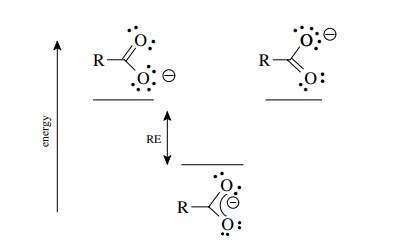
A
molecule for which resonance forms can be written is more stable than any of
the contributing resonance forms. Thus the carboxylate ion (a resonance hybrid)
is more stable than either of the contributing resonance forms. The dif-ference
in energy between the energy of the molecule and the energy of the most stable
resonance form is the resonance energy (RE) of the molecule. The reso-nance
energy represents the stabilization of the molecule due to the delocalization
of electrons.
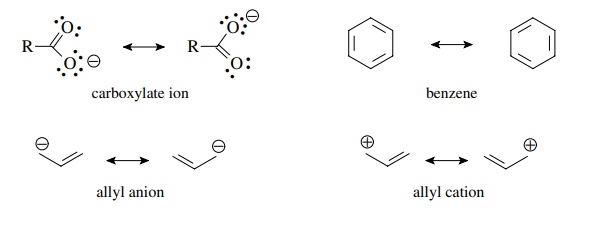
The
amount of resonance energy is related to the relative energies of the
con-tributing resonance forms. The greatest resonance stabilization is found
when the contributing resonance forms are degenerate (equal) in energy. Thus
molecules such as the carboxylate ion, benzene, the allyl anion, and the allyl
cation all have significant resonance stabilization because the main resonance
contributors are of the same energy.
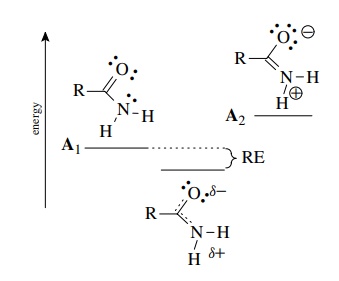
In
contrast, resonance stabilization is less in an amide because the resonance
forms A1 and A2 given below are very
different in energy. Nevertheless, because an amide is a resonance hybrid of A1 and A2, it is predicted that there should be some
double-bond character in the bond between carbon and nitrogen. This is in fact the
case since many amides show restricted rotation around the C–N bond (typical of
a π bond). Moreover, the nitrogen
atom in amides is nearly planar and not very basic, also indicating that the
lone pair is delocalized.
It
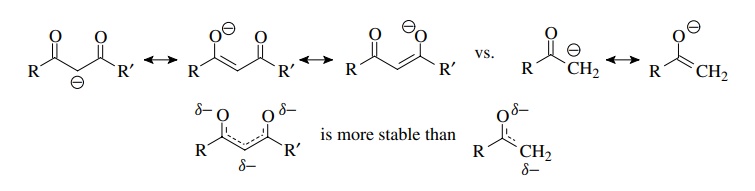
is also generally true that the greater the number of contributing resonance
forms, the greater will be the resonance stabilization. For this reason the
enolate of a β-diketone has much more
resonance stabilization than the enolate of a simple ketone (three resonance
forms versus two). The electrons are delocalized over five atoms in the former versus
three atoms in the latter. In addition, the electron density on the carbon atom
is less in the diketone enolate than in a simple methyl ketone enolate.
Resonance
has a significant influence on the electron distributions and energies of
molecules. The delocalization of electrons is described by the contributions of
resonance forms, which are themselves localized structures with discrete bonds.
Such structures are known as valence bond (VB) structures, and this approach to
the description of bonding in molecules is called the valence bond approach. As
long as one keeps in mind that resonance forms are limiting VB structures and
that the actual molecule is a resonance hybrid of these VB structures, a great
deal of insight into the structure and properties of molecules can be gained.
Related Topics
