The clinical application of cerium salts
| Home | | Inorganic Pharmaceutical Chemistry |Chapter: Essentials of Inorganic Chemistry : The Clinical Use of Lanthanoids
The element cerium has the chemical symbol Ce and atomic number 58. It is a silvery and soft metal, which easily is oxidised in air. Cerium has three oxidation states, +II, +III and +IV, the last being the more stable one.
The
clinical application of cerium salts
The element cerium has the chemical symbol Ce
and atomic number 58. It is a silvery and soft metal, which easily is oxidised
in air. Cerium has three oxidation states, +II, +III and +IV, the last being
the more stable one. The electronic configuration of the resulting Ce4+
ion is [Xe]4f0. Ce+2 is the rarest oxidation state and
the resulting electronic configuration is [Xe]4f2.
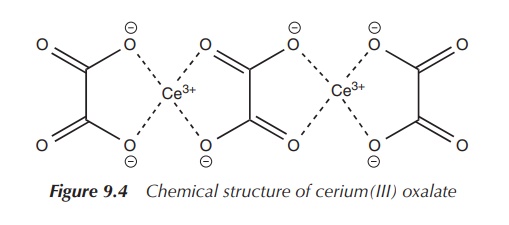
The clinical use of cerium dates back to the mid-nineteenth
century, when cerium(III) oxalate was used as an antiemetic. The exact mode of
action is unknown, but it is believed to be a local effect limited to the GI
tract. This is based on the fact that lanthanoids are not easily absorbed after
oral administration, as previously discussed, and that cerium oxalate has a low
aqueous solubility. Nevertheless, the clinical use of cerium salts as
antiemetic was eventually replaced by antihistamines (Figure 9.4) .
At the end of the nineteenth century, several Ce3+
salts were under investigation for their antibacterial activity in burn wounds.
Especially, cerium nitrate [Ce(NO3)3] showed broad
activity against a variety of pathogens and was subsequently used in
combination with silver sulfadiazine. Initial studies were very suc-cessful and
an estimated reduction of 50% of the mortality rate was suggested. It was
believed that this result was due to the synergistic antimicrobial effect of
both reagents .
Recent studies have shown that cerium nitrate has no significant effect on pathogens from burn wounds. Fur-thermore, research has shown that the suppression of the immune system in patients with serious burn wounds is a main factor for mortality. It has also been shown in animal models that cerium nitrate is a modulator of the burn-associated immune response. Nowadays, this is believed to be the main role of cerium(III) nitrate when used as part of a combination treatment of burn victims. Currently, the cerium salt is used in combination with silver sulfadiazine in individual cases for the treatment of life-threatening burn wounds. Reports suggest that wound healing improves, mortality rates drop and graft rejection rates are also significantly lower .
Related Topics
