Therapeutic use of radiopharmaceuticals
| Home | | Inorganic Pharmaceutical Chemistry |Chapter: Essentials of Inorganic Chemistry : Radioactive Compounds and Their Clinical Application
Radiopharmaceuticals that are used therapeutically are molecules with radiolabelling. This means that certain atoms in this molecule have been exchanged by their radioactive isotopes.
Therapeutic
use of radiopharmaceuticals
Radiopharmaceuticals that are used therapeutically are molecules
with radiolabelling. This means that certain atoms in this molecule have been
exchanged by their radioactive isotopes. These radiolabelled molecules are
designed to deliver therapeutic doses of ionising radiation (mostly
β-radiation) to specific disease sites around the body. The more specific the
targeting is, the fewer the side effects expected. For any design of a treatment
regime including radiopharmaceuticals, it is important to consider what the
decay properties of the radionuclide are and what the clearance route and rate
from nontarget radiosensitive tissue is.
1. 131Iodine: therapy for hyperthyroidism
Iodine has the chemical symbol I and atomic number 53. It is a
member of the halogens (group 17 of the periodic table of elements) (Figure
10.13).
Elemental iodine is characterised by the purple colour of its
vapour. Free iodine typically exists (like the other halogens) as the diatomic
molecule I2. Iodide (I−) is the highly water-soluble
anion, which is mainly found in the oceans. Iodine and its compounds are mainly
used in nutrition. It has relatively low toxicity and is easy to include into
organic compounds, which has led to its application as part of many X-ray
contrast agents. Iodine is required by humans to synthesise the thyroid
hormones, and therefore iodine will accumulate in the thyroid gland. Iodine has
only one stable isotope (12753I), but it has several
radioactive isotopes. Some of these are used for medicinal purposes including
diagnostic tests and treatment. Radioisotopes of iodine will accumulate in the
thyroid gland and therefore can be used clinically. The radioactive isotope 129I
has a half-life of 15.7 million years, 125I has 59 days and 123I
has 13 h. The last one is used in nuclear medicine as an imaging agent because
of its gamma radiation and its short half-life. Using a gamma camera, images of
the human body can be made showing areas of accumulation of the radioisotope.
131I is the product of nuclear fission (as experienced during the Chernobyl disaster) and is a β-emitting radioisotope which will be transported to the thyroid gland if inhaled. Fortunately, it can be replaced by treatment with potassium iodide (nonradioactive), which will replace the radioisotope. Nevertheless, 131I can be used as a therapeutic agent against thyroid cancer when applied in high doses. Paradoxically, the β-emitting radioisotope causes cancer when it is applied in low doses, but it will destroy its surrounding tissue if the dose is high enough. Therefore, preparations containing 131I− are often used to treat hyperthyroidism. These preparations are normally administered orally either as capsules or solution.
2. 89Strontium
Strontium is an alkaline-earth element with the atomic number
38, is a member of group 2 in the periodic table of elements and has the
chemical symbol Sr (Figure 10.14).
Strontium is a soft grey metal and is more reactive with water
than calcium. On contact with water, it pro-duces strontium hydroxide and
hydrogen gas. In order to protect the element, strontium metal is usually kept
under mineral oil to prevent oxidation. Natural strontium is formed of a
mixture of four stable isotopes – 84Sr, 86Sr, 87Sr
and 88Sr, with the last one being the predominant one.
89Sr is
an artificial radioisotope and is a β-emitter with a half-life of 50.5 days. It
is a product of the neutron activation of 88Sr and decays to the
stable 89yttrium. Metastron is a product containing 89Sr
and is licensed by the FDA. It comes in a ready-to-use vial and expires within
28 days. It is supplied with a calibration vial, so that the pharmacist will be
able to ensure that the patients get the accurate dose prescribed .
Because of the similarity of strontium and calcium (neighbouring
elements in the periodic table of ele-ments), strontium is believed to be
metabolised in the human body in a similar way and accumulates, for example, in
the bones. This has led to its application as a treatment option for pain
caused by bone metastasis. It is known that >50% of
patients with prostate, breast or lung cancer will develop painful bone
metastasis. The exact mechanism of relief from bone pain is not known. 89SrCl2
is administered intravenously and, as its distribution in the human body is
similar to that of calcium, it is quickly cleared from the blood and deposited
in the bone mineral. Strontium can be found in the hydroxyapatite cells of the
bones rather in bone marrow cells. The radioisotope 89Sr delivers localised
β-radiation, inducing a pain relieving effect. A majority of the administered
SrCl2 is actively distributed to the metastases. Any free SrCl2
is excreted renally or along with the faeces .
Low platelet count is the most likely side effect occurring in
patients being treated with 89SrCl2. Platelet counts
should return to preadministration levels after 6 months once treatment is
finished. Treatment with 89SrCl2 is not recommended in
patients with an already low platelet or white blood cell count, and for
patients receiving this treatment the blood parameters have to be regularly
checked even after the treatment is completed.
3. Boron neutron capture therapy (BNCT)
Boron has two stable isotopes, 10B and 11B,
and 14 radioisotopes with very short half-lives. 11B is the most
abundant isotope and represents 80% of natural boron, whilst 10B (∼20%) finds a significant clinical appli-cation
in the so-called boron neutron capture therapy (BNCT).
BNCT is a noninvasive treatment option for malignant tumours, especially
brain tumours and head and neck cancers, and is currently under clinical
trials. The patient is injected with a nonradioactive 10B-containing
compound that acts as a neutron-capturing agent and shows high selectivity to
cancer tissues. Once the compound has reached the tumour, the patient is
exposed to a beam of low-energy neutrons, the so-called epithermal neutrons.
These neutrons lose their energy once they penetrate the skin, but they can
still interact with the neutron-capturing agent and initiate a nuclear
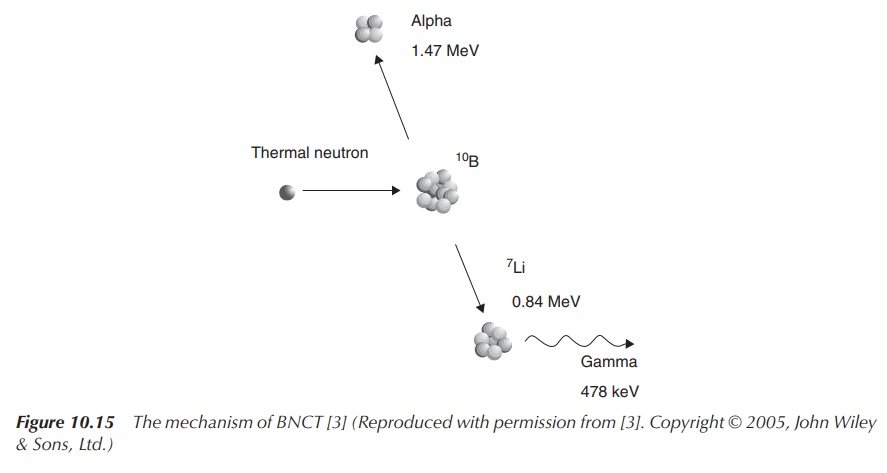
reaction. This reaction of 10B with a neutron results in the
conversion to the nonradioactive isotope 7Li and low-energy gamma
radiation together with the emission of α-radiation (42He2+
particles). α-Radiation is a radiation with a short range and bombards the
local tumour tissue from within the tumour cells. The linear energy transfer
(LET) of these α-particles ranges approximately one cell diameter, which means
there is minimum exposure to healthy tissue (Figure 10.15).
10B + nth
→ [11B]→4He+7Li
+ 2.31 MeV
A variety of carrier molecules for 10B have been
investigated, including carbohydrates, antibodies, liposomes and amino acids.
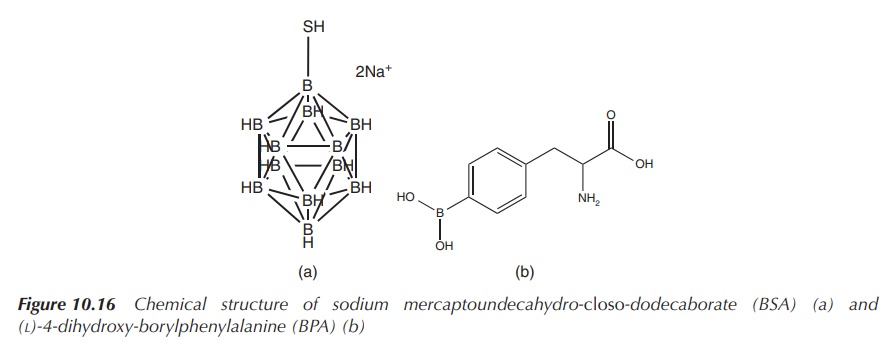
There are currently only two boron compounds as BNCT delivery agents used in
clinical trials. Sodium mercaptoundecahydro-closo-dodecaborate
(Na2B12H11SH), known as borocaptate (BSH), was mainly used in clinical trials in Japan, whereas
the boron-based amino acid (L)-4-dihydroxy-borylphenylalanine
(BPA, boronophenylalanine) is used in clinical trials in Europe and the United
states (Figure 10.16) [3, 4].
Related Topics
