Definitions and architectures of polymers
| Home | | Pharmaceutical Drugs and Dosage | | Pharmaceutical Industrial Management |Chapter: Pharmaceutical Drugs and Dosage: Pharmaceutical polymers
Polymers are high molecular weight natural or synthetic molecules made up of small repeating units, the connected molecular structure that repeats over and over again in a polymer.
Definitions and
architectures of polymers
Polymers
are high molecular weight natural or synthetic molecules made up of small repeating units, the connected molecular
structure that repeats over and over again in a polymer. The structures of
common polymers, their repeating units, and their monomers are shown in Figure 11.1. The structural unit enclosed in brackets
or parentheses is referred to as the repeating
unit. To indicate the repetition, a subscript n is frequently placed after the closing bracket, for example,
-[-CH2CH2-]n-. For polymers of a well-defined
and known number of repeating units, the number of mono-meric units
constituting a polymer replaces the subscript “n.”
Polymers
are synthesized from simple molecules called monomers by a process called polymerization.
The structure and molecular formula of the monomer and the repeating unit are very
similar but not exactly the same.
If
only a few monomer units are joined together, the resulting low molec-ular
weight polymer is called an oligomer.
For example, dimer, trimer, and tetramer are structures formed with two, three,
or four monomer units, respectively.
End groups: There are the
structural units that terminate polymer chains. Where end groups are specified, they are shown outside the
brackets, for example:
CH
3CH 2 −
[ − CH2CH2
−]n − CH 2CH3
Homopolymers are composed of
single, identical repeating units forming
the polymer chain or backbone. Heteropolymers
or heterochain polymers contain more
than one type of repeating unit in their backbone. When two or more monomers
combine in specific repeating pattern to make a hetero-polymer, the polymer is
called a copolymer. In copolymers,
the monomeric units may be distributed randomly (random copolymer), in an
alternating fashion (alternating copolymer), or in blocks (block copolymer). A
graft copolymer consists of one polymer branching from the backbone of the
other. Polymer molecules may be linear or branched, and separate linear or
branched chains may be joined by cross-links.
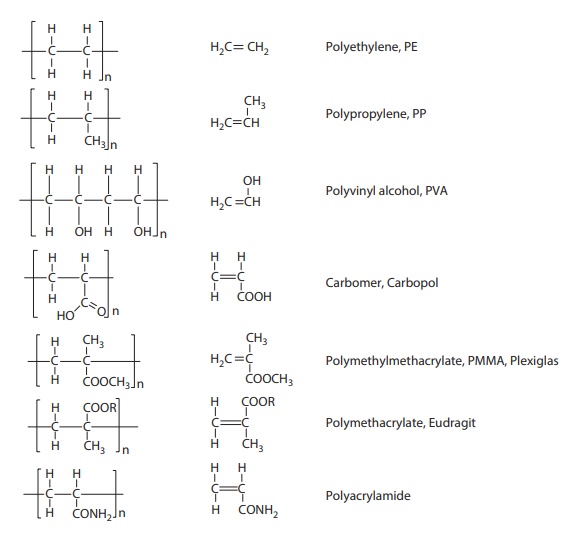
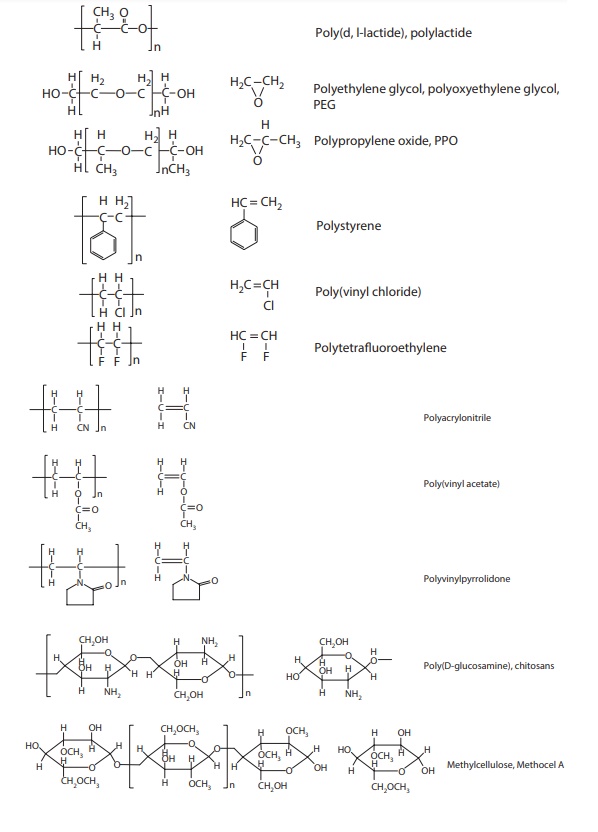
Figure 11.1 Structures of commonly used
polymers and their monomers
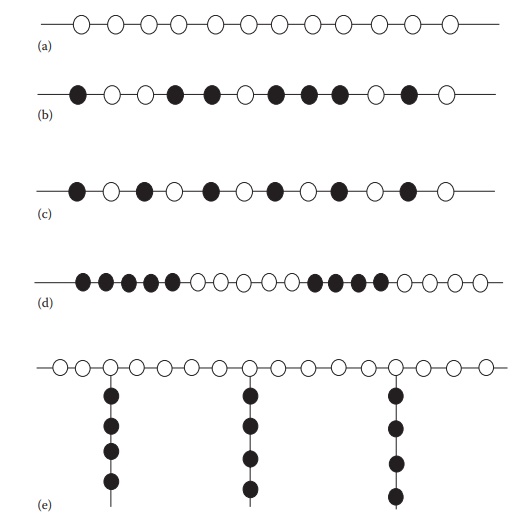
Figure 11.2 Polymer architectures: (a) linear homopolymer, (b) random copolymer, (c)
alternating copolymer, (d) block copolymer, and (e) graft copolymer.
Figure 11.2 shows various arrangements of the hypothetical
monomers A and B in the copolymer. Where blocks of A (○ ) and B (● )
alternate in the backbone, the polymer is designated an -[-AB-]- multiblock copolymer. If the backbone
consists of a single block of each, it is an AB (○●) diblock copolymer. Other possibilities include ABA (○●○) or BAB (●○●)
triblock copolymers. For example,
when vinyl pyrrolidone, a monomer, is polymer-ized, it forms the linear polymer
PVP, also known as povidone. Polyvinyl pyrrolidone is a commonly used polymer
in pharmaceutical processing and products, such as artificial tears. It is a
protective colloid capable of forming complex with molecular iodine and is thus
used in iodine tincture. Polypropylene sulfone is an alternating copoplymer
synthesized by copoly-merization of propylene and sulfur dioxide.
Polymers
can be linear, star-shaped, or branched, including the so-called star block
copolymers. A branched polymer is not necessarily a graft poly-mer. Star polymers contain three or more
polymer chains emanating from
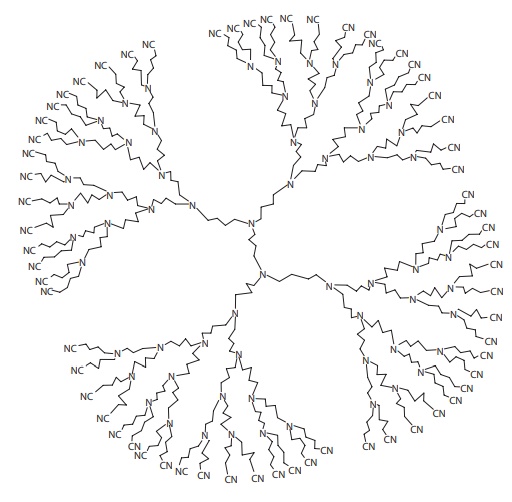
Figure 11.3 Structure of a typical dendrimer polymer.
Comb polymers
contain pendant chains (which may or may not be of equal length) and are
related structurally to graft copoly-mers. Dendrimers,
also known as starburst or cascade polymers, resemble star
polymers, except that each leg of the star exhibits repetitive branching in the
manner of a tree. Dendrimers are highly branched polymer con-structs formed
from a central core, which defines their initial geometry (Figure 11.3). Their branch-like structure leads to a
spherical shape, which can become as large as the size of micelles or
nanospheres, depending on the size of the polymer.
Related Topics
