Micelles
| Home | | Pharmaceutical Drugs and Dosage | | Pharmaceutical Industrial Management |Chapter: Pharmaceutical Drugs and Dosage: Surfactants and micelles
At low concentrations in solutions, amphiphiles exist as monomers and predominantly occupy the surface or interface.
Micelles
At
low concentrations in solutions, amphiphiles exist as monomers and
predominantly occupy the surface or interface. As the concentration is
increased above the level required to completely occupy the surface (known as
the critical micelle concentration or the critical micellization concen-tration
and abbreviated as CMC), subvisible
self-association structures form in solution. These soluble aggregates, which
may contain up to 50 or more monomers, are called micelles. Therefore, micelles are small, gener-ally spherical
structures composed of both hydrophilic and hydrophobic regions of surfactant
molecules. In an aqueous bulk solution environment, the hydrophobic region is
embedded on the inside (Figure 10.3).
Conversely, in a hydrophobic, lipid, or lipophilic bulk solution, the
hydrophilic region is embedded on the inside.
The
surfactant monomers in micelles are in dynamic equilibrium with free molecules
(monomers) in solution, resulting in a continuous flux of monomers between the
solution and the micellar phase.
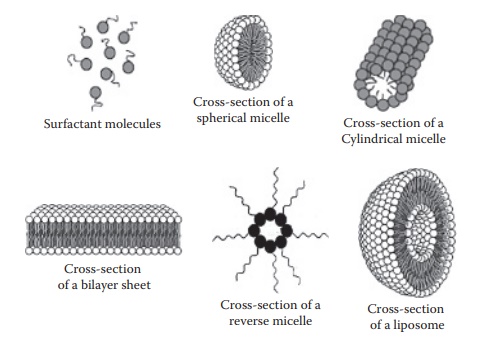
Figure 10.3 Types of micelles. Spherical micelles are formed when the concentration
of monomers in the aqueous solution reaches the critical micelle concentration
(CMC). Elongation of spherical micelles at high concentration leads to the
for-mation of a cylindrical micelle. Reverse micelles are formed in a nonpolar
solvent.
Types of micelles
The
shape of micelles formed by a particular surfactant is greatly influ-enced by
the geometry of the surfactant molecules. At higher surfactant concentrations,
micelles may become asymmetric and eventually assume cylindrical or lamellar structures
(Figure 10.3). Thus, spherical micelles exist at concentrations relatively
close to the CMC. Oil-soluble surfactants have a tendency to self-associate
into reverse micelles in nonpolar
solvents, with their polar groups oriented away from the solvent and toward the
cen-ter, which may also enclose some water (Figure
10.3).
Micelles versus liposomes
Micelles
are unilayer structures of surfactants, whereas liposomes have a lipid bilayer
structure that encloses the solvent medium (water) (Figure
10.3). Although both micelles and liposomes are formed from amphiphilic
mono-mers, the structure and properties of the monomers play a role in
determin-ing which of these structures forms. In addition, liposomes are not
formed spontaneously—they require an input of energy and are typically formed
by the application of one or more of agitation, ultrasonication, heating, and
extrusion.
Colloidal properties of micellar solutions
Micellar
solutions are different from other types of colloidal solutions (such as
colloidal suspensions of particles), since micelles are association colloids; that is, the associated surfactant molecules
are colloidal in size in solution. The micelles are formed by reversible
self-association of monomers. The minimum concentration of a monomer at which
micelles are formed is called the critical
micelle concentration or the critical micellization concentration (CMC).
The
number of monomers that aggregate to form a micelle is known as the aggregation number of the micelle. The
size of micelles depends on the number of monomers per micelle and the size and
molecular shape of the individual monomers. For example, the longer the
hydrophobic chain or the lower the polarity of the polar group, the greater the
tendency for mono-mers to escape from
water to form micelles and, hence, lower the CMC. The CMC and number of
monomers per micelle differ for different types of surfactants. Some examples
are listed in Table 10.4.
As
the surfactant concentration in a solution is progressively increased, the
properties of the solution change gradually. Not all surfactants form micelles.
In the case of surfactants that form micelles, a sharp inflection point in the
physical properties of the solution is observed at the CMC. The properties that
are affected include the following:
Surface tension: As illustrated in Figure 10.4,
surface tension of a surfac-tant solution decreases steadily up to the CMC but
remains constant above
Table 10.4 Critical micellization concentration and number of surfactant molecules
per micelle
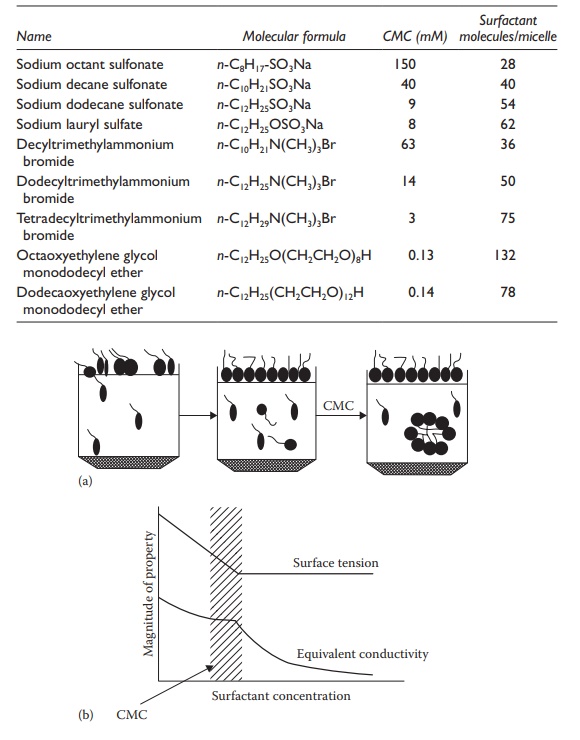
Figure 10.4 Micellization of an ionic surfactant (a) and its effect on conductivity and surface tension (b).
This is attributed to the saturation of surface occupation of a surfactant
above the CMC. Below the CMC, as the surfactant concentration in the solution
is increased, more and more surfactant molecules partition into the surface or
interface, leading to a steady reduction in surface ten-sion. Above the CMC,
the surface or interface is already completely full or saturated with the
surfactant. Thus, further addition of the surfactant leads to minimal changes
in surface tension. The excess surfactant added to the solution forms micelles
in the bulk of the liquid.
Conductivity: The conductivity
of a solution due to the presence of mon-ovalent inorganic ions is affected by
the surfactant’s concentration, since the polar head group of the surfactant
can bind the ions, leading to reduced number of free ions available for
conductance. As a surfactant is added to the solution, some of the surfactant
occupies surface and some is available in the bulk of the solution, binding the
counterions. Thus, solution con-ductivity reduces steadily as a function of the
surfactant’s concentration. As shown in Figure 10.4,
this change is much more rapid above the CMC, following a sharp inflection
point at the CMC. This is attributed to most of the added surfactant (above the
CMC) being available in solution for bind-ing with the ions.
Solubility: Solubility of a
hydrophobic molecule in an aqueous solution
increases slightly with the surfactant concentration below the CMC but
shows significant and sharp increase above the CMC. Below the CMC, an increase
in the solubility of a hydrophobic drug results from changes in the
characteristics of the solvent medium (such as dielectric constant) and
drug–surfactant interaction. Above the CMC, additional drug solubi-lization
results from the hydrophobic drug getting incorporated into the micelles.
Osmotic pressure: Micelles, formed
above the CMC, act as association col-loids, leading to an increase in the
osmotic pressure of the colloidal solution.
Light-scattering
intensity:
Light scattering shows a sharp increase above the CMC due to the formation of colloidal micelles that scatter
light.
Factors affecting critical micelle concentration and micellar size
·
Size and structure
of hydrophobic group: An increase in the hydro-carbon chain length causes a
logarithmic decrease in the CMC. This is because an increase in hydrophobicity
reduces aqueous solubility of the surfactant and increases its partitioning
into the micelles. Micellar size increases with an increase in the hydrocarbon
chain length, owing to an increase in the volume occupied per surfactant in the
micelle.
·
Nature of
hydrophilic group: An increase in hydrophilicity increases the CMC due to increased surfactant
solubility in the aqueous medium and reduced partitioning into the interface.
As the propor-tion of surface/interface to bulk surfactant concentration
reduces, more of added surfactant is required to achieve saturation of the
sur-face before micelles can form. Thus, nonionic surfactants have very lower
hydrophilicity and CMC values compared with ionic surfac-tants with similar
hydrocarbon chains.
·
Nature of
counterions:
About 70%–80% of the counterions of
an ionic surfactant (e.g., Na+
is a counterion for carboxylate and sulfonate groups, and Cl- is a counterion
for quaternary amine groups) are bound to the micelles. The nature of the
counterion influences the properties of these micelles. For example, size of
micelles formed with a cationic surfactant increases according to the series Cl–
< Br– < I– and with an anionic surfactant according
to the series Na+ < K+ < Cs+. This is a
function of not only the size and electronegativity of the counterion but also
the size of the hydration layer around the counterion. The weakly hydrated
(smaller, highly electronegative) ions are adsorbed more closely to the
micellar surface and neutralize the charge on the surfactant more effectively,
leading to the formation of smaller micelles.
·
Addition of
electrolytes:
Addition of electrolytes, such as salt, to solu-tions of ionic surfactants
decreases the CMC and increases the size of the micelles. This is due to a
reduction in the effective charge on the hydrophilic headgroups of the
surfactants. This tips the hydrophilic lipophilic balance toward greater
lipophilicity, increases the propor-tion of surface/interface to bulk surfactant
concentration below the CMC, and promotes the formation of micelles in the bulk
liquid. In contrast, micellar properties of nonionic surfactants are only
mini-mally affected by the addition of electrolytes.
·
Effect of
temperature:
Size of micelles increases and CMC decreases
with increasing temperature up to the cloud point for many nonionic
surfactants due to increased Brownian motion of the monomers. Temperature has
little effect on ionic surfactants. This is due to stron-ger hydrogen bonding
and electrical forces governing the hydrophilic interactions of ionic
surfactants than nonionic surfactants.
·
Alcohol: Addition of
alcohol to an aqueous solution reduces the
dielectric constant and increases the capacity of the solution to
sol-ubilize amphiphilic (surfactant) and hydrophobic molecules. Thus, greater
surfactant solubility in the hydroalcoholic solutions decreases the
surface/interface to bulk solution concentration of the surfactant, thus
increasing the CMC.
Krafft point
Krafft
point (Kt), also known as
the critical micelle temperature or Krafft temperature, is the minimum
temperature at which surfactants form micelles, irrespective of the surfactant
concentration. Below the Krafft point, surfactants maintain their crystalline
molecular orientation form even in an aqueous solution and are not distributed
as freely tumbling random monomers that are able to self-associate to form
micelles. The International Union of Pure and Applied Chemistry’s Gold Book (http://goldbook.iupac. org) defines
Krafft point as the temperature at which the solubility of a surfactant
rises sharply to that at the CMC, the highest concentration of free monomers in
solution. The Krafft point is determined by locating the abrupt change in slope
of a graph of the logarithm of the solubility against temperature (T), or 1/T. Below Kt,
the surfactant has a limited solubility, which is insufficient for
micellization. As the temperature increases, solu-bility increases slowly. At
the Krafft point, surfactant crystals melt and the surfactant molecules are
released in solution as monomers, which can also get incorporated into
micelles. Above the Krafft point, micelles form and, due to their high
solubility, contribute to a dramatic increase in the surfac-tant solubility.
Cloud point
Cloud
point is the temperature at which some
surfactants begin to pre-cipitate and the solution becomes cloudy. The
appearance of turbidity at the cloud point is due to separation of the solution
into two phases. For nonionic surfactants, aqueous solubility is at least
partially attributed to the hydration of their hydrophilic regions by water
molecules. Increasing solution temperatures up to the cloud point leads to an
increase in micellar size. Increasing temperature above the cloud point imparts
sufficient kinetic energy to the hydrating water molecules to effectively
dissociate from the surfactant and bond exclusively with the bulk water. This
produces a suf-ficient overall drop in the solubility of the surfactant to
cause surfactant precipitation and cloudiness of solution. At elevated
temperatures, the sur-factant separates as a precipitate. When in high
concentration, it separates as a gel. This phenomenon is commonly seen with
many nonionic polyoxy-ethylate surfactants in solution.
Organic
solubilized molecules or solution additives, such as ethanol, generally
decrease the cloud point of nonionic surfactants. Addition of aliphatic
hydrocarbons increases the cloud point. Aromatic hydrocarbons or alkanols may
increase or decrease the cloud point, depending on the concentration.
Micellar solubilization
Micelles
can be used to increase the solubility of materials that are normally insoluble
or poorly soluble in the dispersion medium used. This phenom-enon is known as solubilization, and the incorporated
substance is referred to as the solubilizate.
For example, surfactants are often used to increase the solubility of poorly
soluble steroids. The location, distribution, and orienta-tion of solubilized
drugs in the micelles influence the kinetics and extent of drug solubilization.
These parameters are determined by the molecular loca-tion of the interaction
of drugs with the structural elements or functional groups of the surfactant in
the micelles.
1. Factors affecting the extent of solubilization
Factors
affecting the rate and extent of micellar solubilization include the nature of
surfactants, the nature of solubilizates, temperature, and pH.
1. Nature of
surfactants:
Structural characteristics of a surfactant
affect its solubilizing capacity because of its effect on the
solubiliza-tion site within the micelle. In cases where the solubilizate is
located within the core or deep within the micelle structure, the
solubili-zation capacity increases with increase in alkyl chain length. For
example, there was an increase in the solubilizing capacity of a series of
polysorbates for selected barbiturates as the alkyl chain length was increased
from C12 (polysorbate 20) to C18 (polysorbate 80).
An
increase in the alkyl chain length increases the hydrophobicity of the core and
micellar radius, reduces pressure inside the micelle, and increases the
diffusive entry of the hydrophobic drug into the micelle. In addition, the
solubilization of the poorly soluble drug tropicamide increased with increase
in the oxyethylene content of poloxamer. On the other hand, an increase in the
ethylene oxide chain length of a polyoxyethylated nonionic surfactant led to an
increase in the total amount solubilized per mole of surfactant because of the
increasing number of micelles. Thus, the effect of increase in the number of
micelles of the same (smaller) size can be very different than increase in the
size of micelles.
2. Nature of
solubilizate (drug being solubilized): The location of
solu-bilizates in the micelles is closely related to the chemical nature of the
solubilizate. In general, nonpolar, hydrophobic solubilizates are local-ized in
the micellar core. Compounds that have both hydrophobic and hydrophilic regions
are oriented with the hydrophobic group facing or in the core and the
hydrophilic or polar groups facing toward the sur-face. For a hydrophobic drug
solubilized in a micelle core, an increase in the lipophilicity or the
lipophilic region or surface area of the drug leads to solubilization near the
core of the micelle and enhances drug solubility.
Unsaturated
compounds are
generally more soluble than their satu-rated counterparts. Solubilizates that
are located within micellar core tend to increase the size of the micelles.
Micelles become larger not only because their core is enlarged by the
solubilizate but also because the number of surfactant molecules per micelle
increases in an attempt to cover the swollen core.
3. Effect of
temperature:
In general, the amount of the drug solubilized increases with an increase in temperature (Figure 10.5). The effect is particularly pronounced with some
nonionic surfactants, where it is a consequence of an increase in the micellar
size with increasing temperature.
4. Effect of pH: The main effect of
pH on solubilizing ability of non-ionic surfactants is to alter the equilibrium
between ionized and unionized drugs. The overall effect of pH on drug
solubilization is a function of proportion of ionized and unionized forms of
the drug in solution and in micelles, which is determined by (1) the pKa value of the ionizable
functional group(s), (2) the solubility of the ionized and unionized forms in
the solution, and (3) the solubilization capacity of the micelles for the ionized
and union-ized forms. Generally, the unionized form is the more hydrophobic
form and is solubilized to a greater extent in the micelles than the ionized
form.
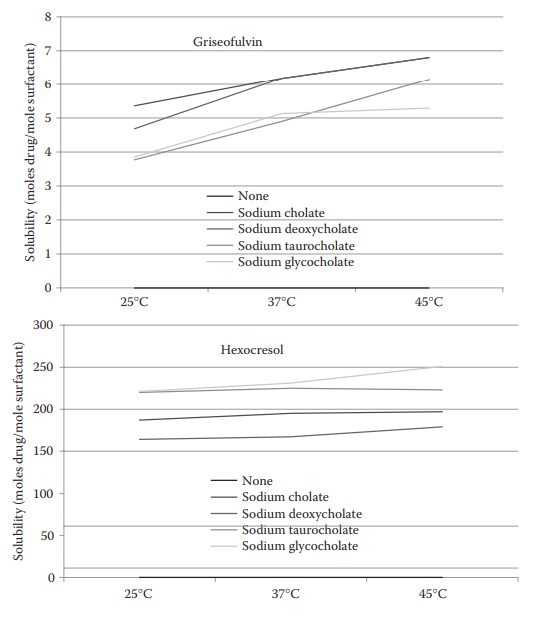
Figure 10.5 Effect of temperature and surfactant type on the micellar
solubilization of griseofulvin and hexocresol. (Modified from Bates, T.R,
Gilbaldi, M. and Kanig, J.I. J. Pharm. Sci., 55, 191, 1966. With Permission.)
2. Pharmaceutical applications
Several
insoluble drugs have been formulated by using micellar solubiliza-tion. For
example:
·
Phenolic compounds, such as cresol, chlorocresol, and
chloroxylenol, are solubilized with soap to form clear solutions for use as
disinfectants.
·
Polysorbates have been used to solubilize steroids in
ophthalmic formulations.
·
Polysorbate are used to prepare aqueous injections of the
water-insoluble vitamins A, D, E, and K.
·
Nonionic surfactants are efficient solubilizers of iodine.
3. Thermodynamics/spontaneity
Micellar
solubilization involves partitioning of the drug between the micel-lar phase
and the aqueous solvent. Thus, the standard free energy of solubi-lization, ∆Gs, can be computed from the
partition coefficient, K, of the drug
between the micelle and the aqueous medium:
∆Gs = −RT In K (10.1)
where:
R is the gas constant
T is the absolute
temperature
Change
in free energy with micellization can be expressed in terms of the change in
enthalpy (∆Hs) and entropy
(∆Ss) as:
∆Gs = ∆Hs − T ∆Ss (10.2)
Thus,
∆H s − T∆ Ss = −RT In K
Or,
In
K = − − ∆Hs/R ⋅ 1/T + constant
where
the constant is ∆Ss/R, assuming that the change in entropy
from micellization is constant. Thus, experimental determination of enthalpy of
micellization can be a useful tool to predict ∆Gs, which, in turn, indicates whether micellar
incorporation of a drug would be spontaneous. When ∆Gs is negative, solubilization process is spontaneous.
When ∆Gs is positive,
solubilization does not occur.
Example 1: Given ∆Hs = 2830 cal/mol and ∆Ss = −26.3 cal/K mol, does
ammonium chloride spontaneously transfer from water to micelles?
∆Gs = ∆Hs − T∆ Ss = 2830 cal/mol − (298K)( − 26.3 cal/kmol)
which
is positive, indicating that micellar solubilization (transfer) would not
occur.
Example 2: Given ∆Hs
= −1700 cal/mol and ∆Ss = 2.1 cal/K mol, does
amobarbital spontaneously transfer from water to a micellar solution (sodium
lauryl sulfate, 0.06 mol/L)?
∆Gs = ∆Hs − T∆ Ss = 1700 cal/mol − (298K)( − 2 .1 cal/kmol) = −2326 cal/mol
which
is negative, indicating that micellar solubilization (transfer) would indeed
spontaneously occur.
Related Topics
