Mechanisms of Action
| Home | | Pharmaceutical Microbiology | | Pharmaceutical Microbiology |Chapter: Pharmaceutical Microbiology : Non-Antibiotic Antimicrobial Agents: Mode Of Action And Resistance
In any consideration of mechanism of action, due regard should be given to the initial health of the organism, duration of contact with the biocide, and the concentration of biocide employed.
MECHANISMS OF ACTION
In any consideration of mechanism of
action, due regard should be given to the initial health of the organism,
duration of contact with the biocide, and the concentration of biocide
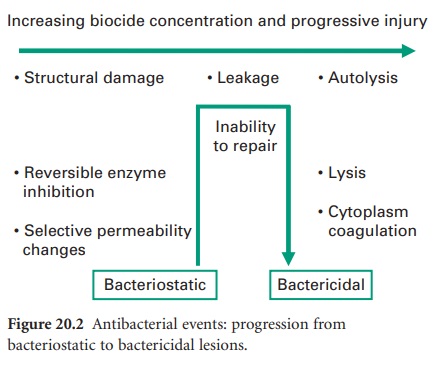
employed. Antibacterial effects may progress from early, sublethal events to
multiple lesions of bactericidal consequence. Figure 20.2 identifies
events in order of severity, but should not be interpreted as defining the
normal progression of cell injury. As disclosed in the following sections, the
biocide interaction may induce particular lesions over others; this will most
certainly be in a concentration-dependant manner
a) Oxidation Reactions
Biocides with oxidizing (electron-withdrawing) ability are widely used
as disinfectants and chemical sterilants, and include the halogens (chlorine,
hypochlorites, bromine, iodophors) and peroxygens (hydrogen peroxide, peracetic
acid and chlorine dioxide). They can exert specific effects on essential
microbial macromolecules causing, variously: strand breakage and adduct
formation on DNA and RNA with disruption of replication, transcription and and
translocation processes; degradation of, particularly, unsaturated fatty acids
leading to loss of membrane fluidity and subsequent reduced functionality of
membrane-bound proteins; and specific modifications to amino acid residues,
most notably disulphide bonds, leading to changes in protein primary structure
and con-formation with consequent disruption of structural enzymic functions.
An accumulation of these effects can be particularly devastating to the
microbial cell.
b) Cross-Linking Reactions
The aldehydes formaldehyde, glutaraldehyde and ortho-phthalaldehyde, and
the sterilant alkylating agents ethylene oxide and propylene oxide, are both
highly reactive chemical classes. The alkylating agents exhibit particularly
strong reactions with guanine residues causing cross-linking between DNA
strands, inhibiting DNA unwinding and RNA translation. The amino, carboxyl,
sulphydryl and hydroxyl groups of structural or enzymic proteins are also
susceptible to alkylation, causing cross-links between adjacent amino acid
chains and also with other amino acid-containing structures such as
peptidoglycan. The aldehydes are generally more specific with greatest effect
against the amino groups of surface exposed lysine or hydroxylysine residues of
proteins, again causing extensive cross-linking.
In all instances, progressive cross-linking leads to macromolecule
malfunction causing inhibition or arrest of essential cell functions. It is
safe to say that there is no single fatal reaction but that death results from
the accumulated effect of many reactions in a manner similar to oxidizing
agents.
c) Coagulation
The cross-linking reactions give rise to
macromolecule denaturation which can be recognized under electron microscopy as
intracellular coagulation. Coagulative effects are not unique to aldehydes and
alkylating agents, however, and high concentrations of disinfectants such as
chlorhexidine, phenol, ethanol and mercuric salts will also coagulate the
cytoplasm. This most likely arises from the precipitation of protein caused by
a variety of interactions including ionic and hydrophobic bonding and the
disruption of hydrogen bonds.
d) Disruption Of Functional Structures
The integrity and functions of the bacterial cell are dependant upon
critical macromolecular structural arrangements including within the cell wall
and cytoplasmic membrane . A number of biocides can have a profound effect on
these organelles.
i) Cell wall
This structure is the traditional
target for a group of antibiotics which includes the penicillins, but a
little-noticed report which appeared in 1948 showed that low concentrations of
disinfectant substances caused cell wall lysis such that a normally turbid
suspension of bacteria became clear. It is thought that these low concentrations
of disinfectant cause enzymes whose normal role is to synthesize the cell wall
to reverse their role in some way and effect its disruption or lysis. In the original
report, these low concentrations of disinfectants (formalin, 0.12%; phenol, 0.32%;
mercuric chloride, 0.0008%; sodium hypochlorite, 0.005% and merthiolate,
0.0004%) caused lysis of Escherichia coli, streptococci,
and staphylococci.
Divalent cations, in addition to their role as enzyme cofactors, also
stabilize cell wall, membrane and ribosomal structures. In particular,
magnesium serves to link the lipopolysaccharide (LPS) of Gramnegative bacteria
to the outer membrane. Chelators, particularly ethylenediamine tetraacetic acid
(EDTA), have been used to disrupt this link and cause the release of LPS into
the medium. The loss of outer membrane integrity and subsequent
permeabilization has been exploited in the potentiation of biocides, including
combinations of EDTA with chloroxylenol, cetrimide, phenylethanol and the
parahydroxy benzoic acid esters.
ii) Cytoplasmic membrane
The bacterial cytoplasmic membrane
consists of an impermeable, negatively-charged, fluid phospholipid bilayer
incorporating an organized array of membrane associated proteins. Through the
membrane-bound electron transport chain aerobically, or the membrane-bound
adenosine triphosphatase (ATPase) anaerobically, the bacterium succeeds in
maintaining a transmembrane gradient of electrical potential and pH such that
the interior of the cell is negative and alkaline. This proton motive force, as it is called, drives a number
of energy-requiring functions which include the synthesis of ATP, the coupling
of oxidative processes to phosphorylation, a metabolic sequence called oxidative phosphorylation, and the transport and
concentration in the cell of metabolites such as sugars and amino acids. This,
put briefly, is the basis of the chemiosmotic theory linking metabolism to
energy-requiring processes.
Certain chemical substances have been
known for many years to uncouple oxidation from phosphorylation and to inhibit
active transport, and for this reason they are named uncoupling agents. They are believed to act by
partitioning into the membrane and rendering it permeable to protons, hence
short-circuiting the potential gradient or protonmotive force. Some examples of
antibacterial agents which owe at least a part of their activity to this ability
are tetra-chloro-salicylanilide (TCS), tricarbanilide, trichlorocarbanilide
(TCC), pentachlorophenol, di-(5-chloro-2-hydroxyphenyl) sulphide (fentichlor), 2-phenoxyethanol,
and lipophilic acids and esters.
The membrane, as well as providing a dynamic link between metabolim and
transport, serves to maintain the pool of metabolites within the cytoplasm. A
general increase in membrane permeability brought about by the association and
likely insertion of biocide molecules into the lipid bilayer was recognized
early as being one effect of many disinfectant substances.
Treatment of bacterial cells with appropriate concentrations of such
substances as cetrimide and other QACs, chlorhexidine, polyhexamethylene
biguanides, phenol and hexylresorcinol causes a leakage of a group of
characteristic chemical species. The potassium ion, being a small entity, is
the first substance to appear when the cytoplasmic membrane is damaged. Amino
acids, purines, pyrimidines and pentoses are examples of other substances which
will leak from treated cells. If the action of the drug is not prolonged or
exerted only in low concentration, the damage may be reversible and leakage may
only induce bacteriostasis. There is however, evidence that a depletion of
intracellular potassium caused by membrane damage can lead to the activation of
latent ribonucleases and the consequent breakdown of RNA. Several biocides,
including cetrimide and some phenols, are known to cause the release of
nucleotides and nucleosides following an autolytic process. This is
irreversible and has been proposed as an autocidal (suicide) process,
committing the injured cell to death (Denyer & Stewart, 1998).
Surface-associated proteins within the membrane fulfil a number of
important roles including wall biosynthesis, nutrient transport and
respiration. Usually enzymes, these macromolecules are often topologically
organized and uniquely exposed to disruption by biocidal agents. Thus,
hexachlorophane inhibits the electron transport chain in bacteria,
chlorhexidine has been shown to inhibit ATPase, and thiol-containing membrane
dehydrogenases are highly susceptible to mercury-containing antibacterials,
silver,2-bromo-2-nitropropan-1,3-diol (bronopol) and isothiazolinones.
Related Topics
