Nonpolymerization Reactions
| Home | | Organic Chemistry |Chapter: Organic Chemistry : Carbon-Carbon Bond Formation By Free-Radical Reactions
The use of free-radical reactions for the preparation of single molecules as products requires greater control of the various steps in the process.
NONPOLYMERIZATION REACTIONS
The use of free-radical reactions for the preparation of single molecules as products requires greater control of the various steps in the process. Traditional free-radical addition reactions carried out in solution (to minimize polymeriza-tion) often gave low yields and mixtures of products and thus were not of real synthetic value. In the ideal case one would want to be able to generate a spe-cific free-radical species which would undergo a single reaction process and then be terminated. The termination step should give a single product plus the reactive radical to continue the chain. Thus very selective methods of initiation are needed, and a clear understanding of propagation and termination steps is required in order to control product formation more closely.
Based
on these requirements for the controlled production of single molecule products
from free-radical reactions, the second phase of free-radical chemistry began
about 20–30 years ago when fast kinetic methods were developed to measure the
rate constants for known free-radical processes in solution. Thus it became
possible to measure the comparative rates of hydrogen abstraction versus olefin
addition and the relative rates of intermolecular versus intramolec-ular
reactions. The results of these investigations led to the realization that
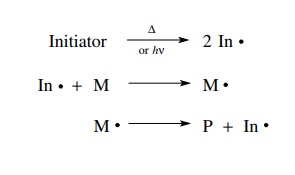
intramolecular olefin additions which produce rings are often much faster than
other reaction pathways, especially in dilute solutions. Moreover, the formation
of five-membered rings by intramolecular olefin addition is much faster than
the formation of rings of other sizes. It is this kinetic selectivity which can
be used as the basis for efficient carbon–carbon bond-forming processes.
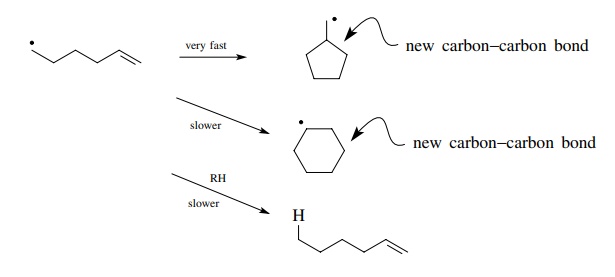
The
identification of this single reaction process which occurs much faster than
others can be used as a focal point for selective carbon–carbon bond
construction. That is, if a radical can be produced on a carbon which is five
carbons away from a double bond, then the fastest reaction which occurs is
cyclization to a five-membered ring. Because free radicals are uncharged,
nonpolar entities, such cyclizations are also found to be largely unaffected by
inductive effects or solvents or substitution patterns. As a consequence
protecting groups are normally not needed for free-radical cyclizations and a
wide range of reaction conditions are compatible with an efficient reaction.
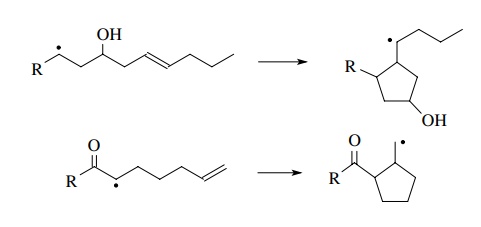
Moreover,
if this cyclization can be incorporated effectively in a chain process, the
cyclized radical would be trapped and yield a single product.
Related Topics
