Free-Radical Reactions
| Home | | Organic Chemistry |Chapter: Organic Chemistry : Carbon-Carbon Bond Formation By Free-Radical Reactions
In such reactions one carbon serves as an electron donor (nucleophile) and a second carbon serves as an electron pair acceptor (electrophile).
CARBON-CARBON BOND FORMATION BY
FREE-RADICAL REACTIONS
FREE-RADICAL REACTIONS
In
the previous chapter the formation of carbon–carbon bonds was discussed in
terms of polar or two-electron processes. In such reactions one carbon serves
as an electron donor (nucleophile) and a second carbon serves as an electron
pair acceptor (electrophile). The result of the donor–acceptor interaction of
these two species is a new carbon–carbon bond in which the electron pair is
shared by the donor and acceptor.
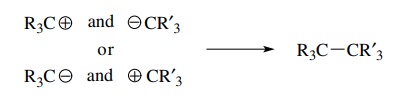
Another
way to form a bond between two carbons is for each carbon atom to supply one
electron. In this case interaction between two carbons which each has a single,
unshared electron would result in formation of a carbon–carbon bond. Species
with unshared electrons are called free radicals, and thus formation of
carbon–carbon bonds by this strategy requires carbon-centered free radicals as
reactants.
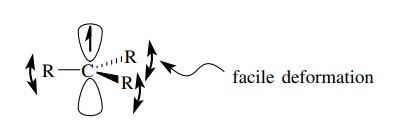
Carbon-centered
free radicals are carbon atoms which have three bonds and seven valence shell
electrons, with the unshared electron occupying a valence orbital. They are
generally thought to be planar (sp2 hybridized) and the unshared
electron is in a 2p atomic orbital. They are not rigidly planar but are easily
deformed to a pyramidal geometry. Because they have only seven valence level
electrons, free radicals are very reactive intermediates and they rapidly
undergo a variety of reactions. Because of their high reactivity, the formation
of bonds by the combination of two free radicals is actually rare because the
free-radical species must survive long enough to encounter another free radical
with which to react. Normally free radicals undergo other reaction processes
before they encounter a second free radical with which they can combine.
The
reactivity of carbon-centered free radicals results from their drive to achieve
an octet electronic configuration, which they do by two principal reaction
processes. The first is atom transfer. This process is one in which an atom
with one electron is transferred from a closed-shell molecule (fully paired,
valence octets) to the free radical.
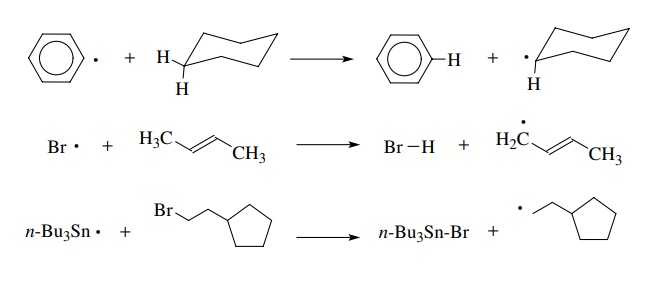
Due
to the conservation of spin, a new radical species is formed. If the atom that
is transferred is a hydrogen, then the process is called hydrogen abstraction
and is the most common atom transfer reaction; however, other atoms can be
transferred to free radicals as well. The driving force for atom transfer
(abstrac-tion) reactions is usually the formation of a stronger bond and/or a
more stable free radical.
A
second very common free-radical reaction is addition to π systems to give a new bond and a new free radical. In this
process the π bond is broken.
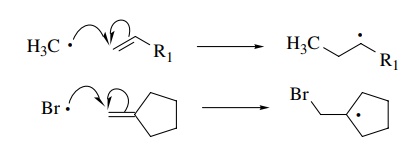
This
process is quite common for carbon-centered free radicals because the
car-bon–carbon σ bond which is formed
is stronger by about 30 kcal than the π
bond which is broken. Other radical species, however, are well known to undergo
olefin additions as well. The addition of bromine to olefins is the key step in
the anti-Markovnikov addition of HBr to olefins.
Another
common feature of free-radical reactions is that they tend to be chain
processes. Since any chemical reaction must exhibit conservation of spin, the
reaction of a free radical with a closed-shell (fully electron paired) molecule
must result in the production of a new free-radical species which can
participate in subsequent free-radical reactions. The series of free-radical
reactions leading to product is often a cyclic process in which the initial
free radical is produced once again in the last step of the cycle so that the
reaction sequence starts over again. The process is termed a chain reaction because each step of the
process is linked directly to the preceding step.
Free-radical
chain reactions can generally be divided into three phases:
1. Initiation is the phase of the
process in which free radicals are produced
that can start the chain reaction.
2. Propagation is the phase of the
process in which free radicals undergo reactions
which form products and produce new free radicals which can continue the chain.
3. Termination is the phase of the
process in which free radicals are removed
from the system by recombination or other reactions, thus interrupting the
chain reaction.
A
classic example is the free-radical addition of chloroform to olefins initiated
by benzoyl peroxide.
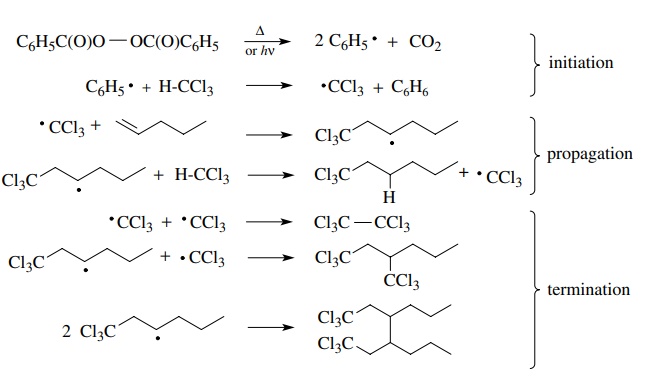
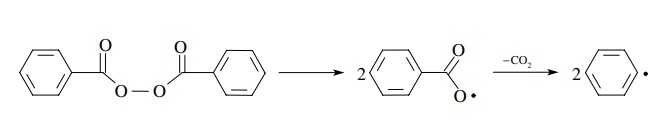
Initiation
normally requires molecules with weak bonds to undergo homolytic cleavage to
produce free radicals. Since bond homolysis even of weak bonds is endothermic,
energy in the form of heat (Δ) or light (hν)
is usually required in the initiation phase. However, some type of initiation
is required to get any free-radical
reaction to proceed. That is, you must first produce free radicals from
closed-shell molecules in order to get free-radical reactions to occur. Benzoyl
peroxide contains a weak O–O bond that undergoes thermal cleavage and
decar-boxylation (probably a concerted process) to produce phenyl radicals
which can initiate free-radical chain reactions.
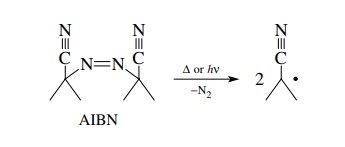
Azobisisobutyronitrile
(AIBN) is perhaps the most widely used initiator. It under-goes either
thermolytic (Δ) or photolytic cleavage (hν)
to give isobutyronitrile radicals which can initiate free-radical reactions.
Hexa-n-butylditin can be photolyzed to two
tri-n-butyltin radicals which are
ini-tiators for tin-based free-radical reactions.
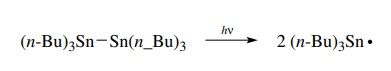
Many
other free-radical initiators are available as well, and the choice of
initiator is normally based on literature precedent and ease of use.
The
propagation phase of a free-radical chain reaction is usually a cyclic sequence
in which a molecule of product is produced and the propagating rad-ical is
regenerated by the sequence. In the above example, the trichloromethyl radical
adds to the double bond to give a new carbon-centered radical which abstracts a
hydrogen from chloroform to produce a molecule of product and another
trichloromethyl radical that continues the chain. Notice that this stage is
cyclic and infinite. If a single ●CCl3 were generated, it would
continue to form one molecule of product and another ●CCl3 until the
olefin was converted com-pletely to product—one molecule at a time. Clearly
this would be very slow, but because a single initiation event can lead to many
molecules of product, one needs very little initiation. If, for example, the
“chain length” of the propagation cycle is 200–300 (a common value), then one
would need only a 1/200–1/300 ratio of initiator to olefin to convert the
olefin completely to product. Thus initi-ation at 0.5–0.3% would suffice. If
chain lengths are longer, then less initiation is required; if they are
shorter, then more initiation would be required.
Termination
reactions, while rare, do occur and serve to interrupt propagation cycles by
removing propagating radicals from the system. Often these reactions are
radical recombinations, but termination reactions also include the reaction of
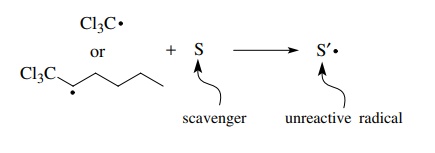
propagating free radicals with other species in solution (called scavengers) to
give radicals incapable of participating in the propagation cycle. In the above
example, a scavenger could react with either the trichloromethyl radical or the
trichloromethyl addition product to give an unreactive free radical and thus
inter-rupt the chain process.
The
more effective a termination step is, the shorter will be the propagation cycle
and the less product will be produced per initiation event. In the limiting
case, if each initiation event was terminated, then no product would be produced. This is the role of “antioxidants”
added to many products and most processed food. These additives scavenge free
radicals produced by the reaction of oxygen with C–H bonds and prevent them
from participating in oxidation propagation cycles—thus oxidative degradation
is stopped or slowed markedly.
It
is the reactivity of free radicals which has made them difficult to understand
and control. Because of their great reactivity, they are quite unselective and
tend to react with anything in solution, and hence multiple pathways and many
prod-ucts are often the rule. Moreover many initiation methods fail to produce
single free-radical species in a controlled and efficient fashion. As a result
of these fac-tors, the use of free radicals in preparative organic chemistry
has seen two distinct phases: first free-radical polymerization and then
nonpolymerization reactions.
Related Topics
