Soft gelatin capsules
| Home | | Pharmaceutical Drugs and Dosage | | Pharmaceutical Industrial Management |Chapter: Pharmaceutical Drugs and Dosage: Capsules
Soft gelatin capsules consist of a hermetically sealed outer shell of gelatin that encloses a liquid or semisolid medicament in the unit dosage.
Soft gelatin
capsules
Soft
gelatin capsules consist of a hermetically sealed outer shell of gelatin that
encloses a liquid or semisolid medicament in the unit dosage. Soft gelatin
capsules are a completely sealed dosage form and cannot be opened without
destroying the capsules. Drugs that are commercially prepared in soft capsules
include cyclosporine, declomycin, chlorotrianisene, digoxin, vitamin A, vitamin
E, and chloral hydrate.
Figure 21.1 shows different shapes of soft gelatin capsules.
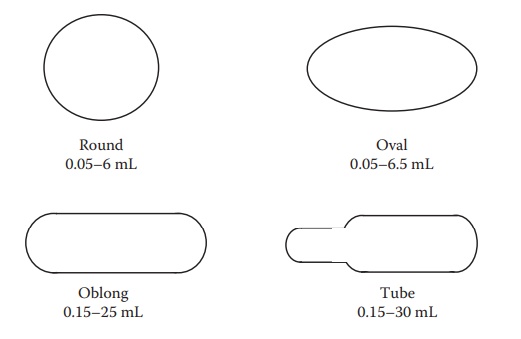
Figure 21.1 Schematic diagrams illustrating different shapes of soft gelatin capsules. The range of fill volumes is also indicated.
Advantages and disadvantages of soft gelatin capsules
Soft
gelatin capsules provide a patient-friendly dosage form for peroral
administration of nonpalatable and/or oily liquids. Solutions or suspensions
with an unpleasant odor or taste can be easily ingested in a soft gelatin
cap-sule dosage form, which offers tidy appearance and convenient ingestion.
This
dosage form can be particularly advantageous for low dose drugs that are lipid
soluble because it can allow greater uniformity of content between dosage units
than the conventional tablet dosage form. It can also be more suitable than a
tablet dosage form for the encapsulation of liquid, water-insoluble drugs. The
capsules can be formulated to be immediate release (IR), slow or sustained
release (SR), or enteric coated.
The
use of soft gelatin capsule shell imposes significant limitations on the drug
formulations that can be encapsulated in this dosage form, that is, restricted
to liquids and semisolids. The manufacturing process is rela-tively tedious and
difficult to optimize (e.g., ribbon thickness, fill weight, and weight
variation). In addition, the breakage of even one capsule during the manufacturing
can lead to the coating of drug formulation on the outer surface of several
other capsules. This can also happen dur-ing storage in multiple use
containers, such as high-density polyethylene (HDPE) bottles.
Soft
gelatin capsules have certain disadvantages compared to liquid-filled hard
gelatin capsules. Due to the relatively higher water content in soft gela-tin
shell (20–30% w/w) compared to hard gelatin capsules (13–16% w/w)
moisture-sensitive drugs may not be stable in soft gelatin capsules. In addi-tion,
the maximum temperature of the formulation that can be filled into soft gelatin
capsule without deformation of the shell and other production issues is about
35°C, whereas a formulation can be filled at up to 70°C in hard gelatin
capsules without shell deformation. Extreme acidic and basic pH must also be
avoided because a pH below 2.5 hydrolyzes gelatin, whereas a pH above 9 has a
tanning effect on the gelatin.
Drivers for development of soft gelatin capsules
Soft
gelatin capsules are often developed for one or more of the following reasons:
1. Line extension products for
strategic marketing advantage in a thera-peutic area with intense competition.
For example, cough and cold medicines available as a soft gelatin capsule can
offer patient benefit, such as ingestion without water and portability.
2. Technological
advantage such
as good content uniformity of a low dose
drug or formulation of a water-insoluble drug that is liquid at room
temperature.
3. Safety reasons during
product manufacturing, dispensing, and usage. For example, most of the product manufacturing unit operations of
tablets and hard gelatin capsules involve handling of fine powders. In the case
of soft gelatin capsules, the powder handling is restricted to drug dissolution
or dispersion in a liquid medium. Powders inher-ently have greater exposure
hazards than liquids. Therefore, soft gel-atin capsules provide greater
operator safety during manufacturing. In addition, as the drug formulation is
hermetically sealed in a shell, the exposure to the medication is minimized
during dispensing as well as use.
4. Improved oral
bioavailability: The use of certain lipids can be asso-ciated with
increased oral bioavailability and reduced intra- and interpatient variability
by modification of GI digestive processes. In addition, presentation of the
drug in a predissolved state can lead to shorter duration to the onset of
action. By formulating nifedipine or ibuprofen into soft gelatin capsules after
being dissolved in PEG, the bioavailability of these drugs can be improved.
Formulation of soft gelatin capsule shell
The
composition of the soft capsule shell consists of three main ingredi-ents: (1)
gelatin, (2) plasticizer, and (3) water. In contrast to hard gelatin capsules,
a relatively large amount (~30 % w/w) of plasticizers is added in soft gelatin
capsule shell formulation to ensure adequate flexibility. Water is used to form
the capsule, and other additives are often added as needed.
Table 21.3 Typical composition of a soft gelatin capsule shell
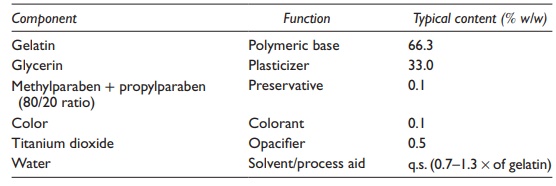
A
typical composition of the soft gelatin capsule shell is listed in Table 21.3 and the functional components are
described as follows:
1. Gelatin: Similar to hard
gelatin shells, the basic component of soft
gelatin shell is gelatin. The properties of gelatin shells are controlled
by the choice of gelatin grade and by adjusting the concentration of
plasticizer in the shell. The physicochemical properties of gelatin are
controlled to allow
·
Adequate flow at desired temperatures to form ribbons of
defined thickness, texture, mechanical strength, and elasticity.
·
Ribbons to be easily removed from the drums, stretch during
fill-ing, seal the temperature below the melting point of the film, and dry
quickly under ambient conditions to an adequate and a repro-ducible strength.
Physicochemical
properties of gelatin important to capsule formation include gel strength,
viscosity, change in viscosity with temperature and shear, melting point,
settling point (temperature), settling time, particle size (affects time to
dissolve), and molecular weight distribu-tion (affects viscosity and strength).
2. Plasticizer: A plasticizer
interacts with gelatin chains to reduce the
glass transition temperature (Tg)
of the gelatin shell and/or promotes the retention of moisture
(hygroscopicity). The most common plasti-cizer used for soft gelatin capsules
is glycerol. Sorbitol, maltitol, and polypropylene glycol can also be used in
combination with glycerol. Glycerol derives its plasticizing ability primarily
from its direct inter-actions with gelatin. In contrast, sorbitol is an
indirect plasticizer because it primarily acts as a moisture retentive agent.
Compared to hard gelatin capsules and tablet film coatings, a relatively large
amount (~30% w/w) of plasticizers are added in a soft gelatin capsule
formulation to ensure adequate flexibility.
3. Water: The desirable
water content of the gelatin solution used to pro-duce a soft gelatin capsule
shell depends on the viscosity of the specific grade of gelatin used. It
usually ranges between 0.7 and 1.3 parts of water to each part of dry gelatin.
After the capsule is formed, most of the water is removed by drying. The
finished soft gelatin capsules contain 13–16 % w/w water.
4. Preservative: Preservatives are
often added to prevent the growth of bacteria
and mold in the gelatin solution during storage. Potassium sorbate, and methyl,
ethyl, and propyl hydroxybenzoate are com-monly used as preservatives.
5. Colorant and/or
opacifier:
A colorant and/or opacifier (e.g., titanium
dioxide) may be added to the shell for visual appeal and/or reducing the
penetration of light for the encapsulation of a photosensitive drug. The color
of the capsule shell is generally chosen to be darker than that of its
contents.
6. Other excipients: Other,
infrequently, used excipients can include fla-vors and sweeteners to improve
palatability and acid-resistant poly-mers to impart enteric release
characteristics. They can also be used to formulate chewable soft gelatin
capsules, for example, ChildLife’s Pure DHA chewable 250 mg soft gel caps. A
chelating agent, such as ethylene diamine tetracetic acid (EDTA), can be added
to prevent chemical degradation of oxidation sensitive drugs catalyzed by free
metals in gelatin, such as iron.
Drug formulation for encapsulation in soft gelatin capsules
Soft
gelatin capsules may contain a liquid or semisolid solution, suspension, or
preconcentrate of a self-emulsifying or self-microemulsifying system. For
example, Accutane® is a suspension of isotretinoin in oil, Sandimmune® is a self-emulsifying
preconcentrate, and Neoral® is a self-microemulsifying preconcentrate.
Formulation
considerations for the contents of the soft gelatin capsules include the
following:
·
Noninteraction with
gelatin:
The contents of the soft gelatin capsule should
not interact with the gelatin shell.
·
Nonmoisture
sensitivity:
The moisture content of soft gelatin cap-sules plasticized with glycerol is
considerably higher than that of hard gelatin capsules. Therefore, to ensure
chemical stability of the drug, moisture-sensitive drugs should not be
formulated in soft gelatin capsules.
·
Nontemperature
sensitivity:
The molten gelatin mass usually has a pourable
viscosity at 60°C–70°C. Therefore, the sealing operation is usually carried out
at a higher than ambient temperature. Hence, highly thermolabile drugs may not
be encapsulated in soft gelatin capsules.
·
pH: Extreme acidic and
basic pH should be avoided because a pH below
2.5 hydrolyzes gelatin (leading to leakage), whereas a pH above 9 has a tanning effect on the gelatin. Tanning
process involves cross-linking of gelatin, which results in hardening of the
shell. The shell becomes insoluble in water and resistant to digestion by GI
enzymes: trypsin and chymotrypsin.
Drugs
for encapsulation in a soft gelatin capsule are usually dissolved or suspended
in a suitable carrier. Insoluble drugs are often dispersed or sus-pended in an
agent such as beeswax, soybean oil, or paraffin. Surfactants are often added to
promote wetting of the ingredients. The use of water or ethanol in the fill
composition is only possible with special modifica-tions of the capsule shell.
Drugs can be dispersed in ethylcellulose for an SR effect.
Manufacturing process
Soft
gelatin capsules are filled with solutions or suspensions of drugs in liquids,
and sealed in a single operation. They are prepared from a more flexible
plasticized gelatin by a rotary-die process. As shown in Figure 21.4, this process involves the following
sequential operations:
1.
Two heated sheets of gelatin of similar thickness are
produced by the controlled flow of the fluid gelatin from its heated storage
container (gelatin tank) by using a controlled pore opening and fill in a spreader box.
2.
The gelatin film flows through a series of oil rolls that stretch the sheets and
direct them appropriately toward die
rollers.
3.
The two sheets of gelatin merge on the metallic rollers that
contain dies of appropriate shape and size and move in the opposite direc-tion
toward each other. The application of vacuum inside the rollers combined with
pressurized filling of the components enables the for-mation of a cavity. The
application of heat and mechanical pressure enables sealing of the shells as
they pass through the rollers.
4.
As the gelatin sheets are being annealed, a calibrated
amount of the drug formulation is pumped into each cavity by the product pump through an injection wedge.
5.
The concurrent process of drug product injection into the
die cavity and sealing of the cavity is either accompanied by the cutting and
release of individual soft gelatin capsules (if the rollers are suitably
designed) or, as shown in Figure 21.4, the
capsules may be cut from the sheets in a separate, subsequent operation.
6.
The filled capsules are dried at ambient conditions to
remove moisture from the outer surface and may be tray dried for an extended
period of time (e.g., up to 48 hours).
7. Finished capsules are passed on a conveyor belt for the next unit operations of packaging and labeling.
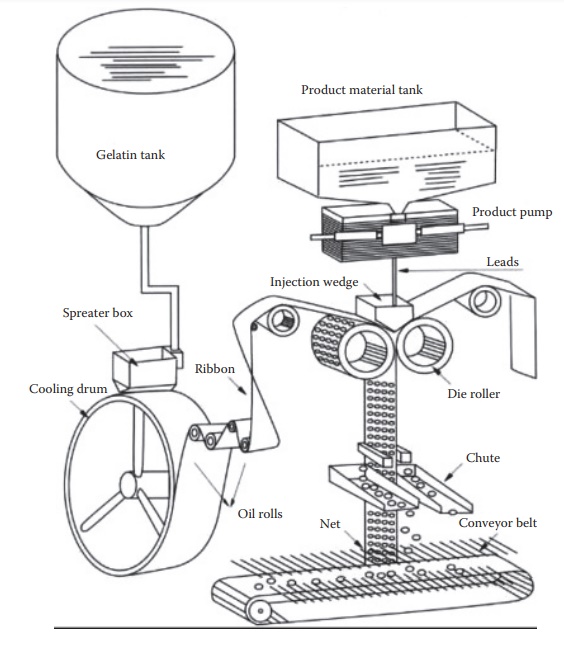
Figure
21.4 Manufacturing process of soft gelatin capsules. (Adapted from http:// www.sunkingpm.com/htm/PM/SCP/5.html)
Nongelatin soft capsules
The
use of alternate polymers for the formation of soft capsules is driven by
marketing or formulation requirements. For example, Vegicaps® are animal-free.
Their shell is made from seaweed extract and gluten-free starch. For moisture
sensitive drugs, HPMC capsules may be preferred, which generally have lower
equilibrium moisture content than gelatin cap-sules. HPMC capsules also have
better physical stability on exposure to low humidity.
Related Topics
