Solubility
| Home | | Pharmaceutical Drugs and Dosage | | Pharmaceutical Industrial Management |Chapter: Pharmaceutical Drugs and Dosage: Pharmaceutical solutions
The required concentration of API in an aqueous solution is determined by the drug’s dose and reasonable amount of solution that can be administered.
Solubility
The
required concentration of API in an aqueous solution is determined by the
drug’s dose and reasonable amount of solution that can be admin-istered. In
addition, factors such as drug’s solubility and taste play a role in
determining drug concentration. For example, the taste of bitter or unpleasant
drugs tends to be concentration dependent. In addition, taste-masking
strategies, such as drug adsorption to ion-exchange resin, limit the maximum
drug concentration in solution depending on the maximum amount of drug that can
be adsorbed on the resin and resin concentration in solution.
Solubilization
of the API, that is, increasing the soluble concentration of the API in the
vehicle, is frequently required to prepare aqueous solutions. The most commonly
used approaches for solubilizing API are the use of one or more of pH control,
surfactant(s), and/or cosolvent(s). Drugs that are poorly soluble in water may
be dissolved in a mixture of water and a water-miscible solvent such as
alcohol, glycerol, polyethylene glycol, or propylene glycol. The proper
selection of a solvent depends on the physicochemical characteristics of the
solute and the solvent.
Temperature
is an important factor in determining the solubility of a drug and in preparing
its solution. Sometimes the manufacturing process requires the use of elevated
temperature to prepare a solution. After manu-facturing, the solution can be
brought back to room temperature without drug precipitation or crystallization.
pH and buffer capacity
The
pH of the vehicle is an important determinant of solubility of an ion-izable
drug. Most drugs are weak acids (e.g., having a carboxylate group) or weak
bases (e.g., having an amine group). Weak acids are ionized at basic pH. Weak
bases are ionized at acidic pH. Ionized forms of the drugs are more soluble
than unionized forms. Thus, pH affects the solubility of the drug. Depending on
the slope of the pH-solubility profile of a drug, a slight increase or decrease
in pH can cause some drugs to precipitate from a solution. Therefore, an adjustment
of pH can aid in solubilizing ionizable drugs, and use of buffer to prevent pH
shift on storage can minimize the risk of precipitation or crystallization.
Buffers
are binary mixtures of compounds in solution that resist changes in solution pH
upon the addition of small quantities of acid or base. These binary mixtures
could be (a) a combination of a weak acid and its conjugate base (i.e., its
salt) or (b) a combination of a weak
base and its conjugate acid (i.e., its salt). A weak acid buffer is exemplified
by the combination of acetic acid and sodium hydroxide, which forms the salt
sodium acetate. A weak base buffer is exemplified by histamine and hydrochloric
acid, which forms protonated histamine chloride salt. Buffer solutions are
generally not prepared from weak bases and their salts because bases are
usually highly volatile and unstable.
pH and buffering capacity
The
most important characteristics of a buffer solution are its pH, which can be
calculated using the Henderson–Hasselbach equation, and its buffer capacity,
which is defined as the magnitude of the resistance of a buffer to pH changes.
The stable pH of the solution generated by a buffer depends on the
concentration of the two species and the pKa
of the weak acid or the weak base. It is determined by the Henderson–
Hasselbalch equation.
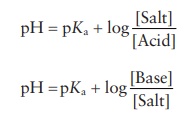
The
extent to which a buffer resists change in solution pH is known as the
buffering capacity. Buffering capacity of a buffer is related to the
concentra-tion of the acid and the base, that is higher the concentration,
greater the buffering capacity. Buffering capacity is generally expressed as
the concen-tration of the buffer. Thus, a 2 M acetate buffer has 10× more
buffering capacity than a 0.2 M acetate buffer. The ratio of the acetate salt
to the acetic acid may be the same in both buffers.
If
strong acid, such as 0.1N HCl, is added to a 0.02 M solution contain-ing equal
amounts of acetic acid and sodium acetate, the pH is changed only 0.09 pH units
because the base acetate (Ac−) ties up the hydrogen ions according
to the reaction:
Ac− + H 3O+ ↔ HAc + H2O
If
strong base, such as 0.1 N NaOH, is added to the buffer mixture, acetic acid
neutralizes the hydroxyl ions as follows:
HAc
+ OH− ↔ H 2O + Ac−
Example of How to Make a Buffer
BUFFERS OF WEAK ACIDS
An
acetate buffer is created by the addition of sodium acetate to acetic acid.
Alternatively, sodium hydroxide can be added to a solution of acetic acid. In
the presence of the strong base, sodium hydroxide, an equimolar amount of acetic
acid, converts to the sodium acetate salt or the acetate ion in situ.
When
sodium acetate is added to acetic acid, the dissociation con-stant, Ka, for the weak acid is
expressed by the equation:
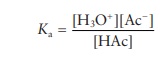
The
dissociation constant is a known constant for each acid. The pKa of acetic acid is 4.75. This means that an equal concentration of acetic acid and sodium acetate in
solution will result in a solution pH of 4.75.
The
pH of the final solution is obtained by rearranging the equilib-rium expression
for acetic acid:
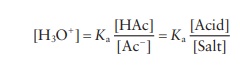
The
aforementioned equation can be expressed in logarithmic form, with the sign
reversed as follows:
log
[H3O+] =
− logKa – log [Acid] + log [Salt]
This
is the Henderson–Hasselbalch equation for a weak acid:
pH
= pKa + log [Salt]/ [Acid]
The
term, pKa, is the negative
logarithm of Ka, which is
called the dis-sociation constant.
BUFFERS OF WEAK BASES
The
buffer equation for solutions of weak base and their salts can be derived in a
manner similar to that for the weak acid buffers. Accordingly,
[OH− ] = pKa [Salt]/[Base]
Using
the relationship [OH−] = Kw/[H3O+],
we can obtain the follow-ing buffer equation:
pH
= pKw − pKb +
log [Salt]/[Base]
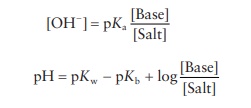
Surfactants and cosolvents
Surfactants
are commonly used in the dosage form to impart an amphiphi-lic character to the
aqueous vehicle and/or associate with the hydrophobic drug to increase its
solubility. When low concentrations of surfactants are added to the aqueous
solution, they associate with the hydrophobic parts of a solute and increase
the solubility of the solute in a concentration-dependent manner. At a certain
concentration, known as the critical micelle concentra-tion (CMC), there are
enough surfactant molecules in solution that several surfactant molecules
self-associate, with hydrophobic parts of the molecule buried inside and the
hydrophilic part on the outside, facing the aqueous environment, to make
structures known as micelles. Typical micelles contain 6–12 molecules of the
surfactant. Micelles are subvisible soluble colloidal structures with a
hydrophobic core. This allows the partition and retention of hydrophobic drug
in the core of the micelle, thus dramatically increasing total drug solubility.
The slope of concentration dependence of solubilization of a solute by a
surfactant is significantly higher above the CMC than below.
Cosolvents
increase drug solubility by altering the dielectric constant and hydrogen bonding
capability of the vehicle and by providing a hydrophobic microenvironment.
Commonly used cosolvents include ethanol, polyethyl-ene glycol, and propylene
glycol. In addition, cyclic polysaccharides, such as cyclodextrins, that have a
hydrophobic cavity and a hydrophilic exterior are often used for drug
solubilization.
Related Topics
