Clinical dose calculations
| Home | | Pharmaceutical Drugs and Dosage | | Pharmaceutical Industrial Management |Chapter: Pharmaceutical Drugs and Dosage: Pharmacy math and statistics
The dose of a drug represents the amount of the drug substance that a patient must take at one time.
Clinical dose
calculations
The
dose of a drug represents the amount of the drug substance that a patient must
take at one time. This amount is designed with an expectation of producing the
optimum therapeutic effect while minimizing the unwanted side effects. In the
current pharmacokinetic paradigm, the designed therapeutic dose for a patient
is usually based on the desired target concentration of the drug substance in
the patient’s central compartment body fluids, that is, blood, or the target
site of action.
Any
changes in the patient’s profile or pathophysiological status that may affect
the drug’s pharmacokinetics can change the drug’s concentration reached in the
patient’s body fluids for the same dose. These changes, for example, can
include patient-to-patient differences in body weight, body surface area (BSA),
age, and renal function. The usual adult dose mentioned for most medications
reflects the amount of drug required for an average 180-lb adult with normal
body functions. The drug’s dose for an individual patient is often adjusted to
reflect one more of these differences, so as to optimize the patient’s exposure
to the drug substance.
Dosage adjustment based on body weight or surface area
In
many cases, the target dose is expressed in terms of BSA or body weight. For
example, meperidine hydrochloride (Demerol®) has a dose of 6 mg/ kg/day in divided
doses to be taken 4–6 times daily, while isoniazid has a recommended daily dose
of 450 mg/m2 BSA/day to be administered in a single dose. Therefore,
the daily dose is calculated based on the patient’s weight or BSA and divided
by the number of doses per day to determine an individual dose amount. A set of
doses administered over a period of time as a part of a treatment plan is
termed dosage regimen.
For
example, for a patient of 180-lb body weight, the daily dose of meperidine
hydrochloride would be 220/2.2 × 6 = 600 mg. For a patient recommended q.i.d.
(Latin, quaque in die, four times a day) dosing for 3 days, the dose would be
600/4 = 150 mg/dose. Therefore, the patient may take three tablets of 50 mg
four times a day. The total number of tablets to be dispensed would be = 3 × 4
× 3 = 36 tablets.
Dosage
calculation based on the BSA is often utilized for the IV admin-istration of
drugs and fluids. The BSA can be calculated by Mosteller’s formula, using the
body weight and height information as follows:

Another, more common, approach to the estimation of BSA is the use of a nomogram (graphical calculation device). Figure 5.1 illustrates a typi-cal adult nomogram. To estimate the surface area, use a ruler to mark the patient’s height and weight in his or her respective scales in a straight line. The point at which this straight line intersects the surface area line is the BSA of the patient.

Figure 5.1 Example of a typical adult nomogram for the calculation of body surface area for patients weighing more than 65 lbs or 3-feet tall. (From http:// www.smm.org.)
Calculation of children’s dose
In
addition to the height and the weight, the BSA is also a function of the age
and gender of an individual. For example, the average BSA of an adult men (~1.9
m2) is higher than that of an adult women (~1.6 m2) and
children (~1.1 – 1.3 m2 for 9- to 13-year-old children). An average
adult’s (150–154 lbs) BSA is assumed to be 1.73 m2. This is often
used in the calculation of children’s doses. For example,
Child’s
dose = Adult dose ( Child’s BSA in m2 / 1.73 m2
)
(5.6)
Estimation
of BSA for children uses a different nomogram, illustrated in Figure 5.2.
Less frequently, a child’s dose is also calculated using the age of child in months (Fried’s rule) or years (Young’s rule) or using the weight of the child in pounds (Clark’s rule). The formulas are illustrated as follows:

Figure 5.2 Example of a typical child
nomogram for the calculation of body surface area for patients weighing less
than 65 lbs or 3 feet tall. (From http://www.smm.org.)
Child’s
dose = Adult dose ( Child’s
weight in pounds / 150 pounds )
The
choice of a formula for dose calculation depends on the conventional practice
of the pharmacy or hospital for a given drug. Attention should also be paid to
the overall metabolic status of the patient and the thera-peutic index of the
drug. For drugs eliminated by the kidney, the renal function, measured by
creatinine clearance (Cr Cl), plays an important role in dose adjustment of
potent compounds. Creatinine clearance of greater than 80 mL/min is considered
normal. For compromised Cr Cl, the formularies usually have a recommended table
of doses, depending on the therapeutic index of the drug and the percentage of
drug eliminated by the kidney.
Dose adjustment for toxic compounds
For
the administration of highly toxic compounds with a narrow thera-peutic window,
such as the cytotoxic anticancer compounds, dosage cal-culation becomes very
critical. These compounds are dosed at very high levels, close to but lower
than their maximum tolerated dose (MTD), to maximize their therapeutic benefit to
the patient. Therefore, interpatient variability in drug exposure has serious
implications on drug effectiveness and toxicity to the patients. The variation
in drug exposure arises from differences in drug metabolism and elimination.
For example, the total body clearance of carboplatin can range from 20 to 200
mL/min owing to interpatient differences in renal function, since most of the
drug is elimi-nated by glomerular filtration. Similarly, topotecan clearance
correlates with renal function.
Different dosage adjustment strategies are followed in these cases, depending on the drug being administered. For drugs with clinically estab-lished exposure–physiological parameter correlations, dosage adjustment for an individual patient is done a priori, based on the patient’s physiolog-ical parameters, such as genotype and/or phenotype of the metabolizing enzymes, renal clearance, serum protein, or hepatic function. In addition, for drugs that are dosed repeatedly or continuously, dosage modification can be based on the measurement of blood levels of the drug and toxici-ties in the patient, for example, for etoposide and fluorouracil.5 Another dose individualization strategy involves administration of a low test dose of the compound to determine the exact pharmacokinetic parameters for an individual patient, followed by modifying the dose to achieve a target drug exposure. In other cases, clinical oncologists frequently use the BSA for drug dose scaling between individuals. Other physiological scaling parameters, such as age, gender, weight, and body mass index, are also used in specific circumstances.
Dose adjustment based on creatinine clearance
Renal function is often determined in terms of a patient’s Cr Cl. Creatinine is a cyclic derivative of the nitrogenous organic acid, creatine (Figure 5.3), found in the muscle. Creatinine is eliminated by filtration through the kid-neys and is not reabsorbed. Therefore, the correlation of its blood and urine levels is an indication of the rate of filtration of blood plasma through the kidney (glomerular filtration rate [GFR]), which indicates renal function. Glomerular filtration rate can be calculated using the concentration of a chemical, such as inulin, that is freely filtered through the kidney but not secreted or reabsorbed.
GFR
= (Urine
concentration × Urine flow ) /Plasma
concentration
The
use of creatinine is preferred over inulin, since extraneous administra-tion is
not required for creatinine. However, a small amount of creatinine is also
secreted by peritubular capillaries, which can contribute to some error
(overestimation) in the calculation of Cr Cl. However, this error becomes
significant only in the cases of severe renal dysfunction.
Creatinine
clearance is estimated by determining blood creatinine con-centration (which is
relatively steady) and the amount of creatinine secreted in urine collected
over a period of 24 h. For example, if 2 mg/mL of creati-nine is detected in 1
L of urine collected over a period of 24 h and the blood creatinine
concentration is 0.01 mg/mL, then:
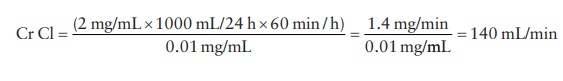
This
is indicative of the rate of filtration of plasma volume through the kidneys
per unit time. Creatinine clearance is often also corrected for the BSA to
normalize dose calculation. Assuming 1.73 m2 as the average-sized
man’s BSA, Cr Cl is expressed as:
Cr
Cl (corrected) = Cr Cl × (1.73/BSA) mL/min/1.73m2
Creatinine
clearance estimation requires and assumes complete urine col-lection over a 24
h period. To avoid this assumption for outpatients, creati-nine clearance can
be estimated on the basis of serum creatinine level alone. For example,
Cockcroft–Gault formula estimates creatinine clearance as:
Cr
Cl = { (140 − age) × weight(kg)×[0.85,if female] } / {72 × serum creatinine level(mg/dL)}
The
normal range of GFR is 100–130 mL/min/1.73 m2. It varies with age,
race, and kidney function. Glomerular filtration rate correlates with different
stages of chronic kidney disease (CKD) as follows:
Stage
1 CKD—GFR greater than 90 mL/min/1.73 m2: normal
Stage
2 CKD—GFR 60–89 mL/min/1.73 m2: mild
Stage
3 CKD—GFR 30–59 mL/min/1.73 m2: moderate
Stage
4 CKD—GFR 15–29 mL/min/1.73 m2: severe
Stage
5 CKD—GFR less than 15 mL/min/1.73 m2: kidney failure
Dose
adjustment based on Cr Cl are provided for most drugs by the manu-facturers
based on the results of clinical trials. These are mainly based on the
percentage of drug eliminated by the kidneys. For highly toxic compounds, Cr Cl
is utilized for the calculation of pharmacokinetic parameter, such as the
elimination rate constant, which is then used with the drug’s pharmacokinetic
model for dose calculation.
The
dosage regimen for a renal-compromised patient is usually adjusted by either reducing
the dose or prolonging the dosing interval. Reduction in dose is recommended
for cases where relatively constant blood level is desired, for example, β-lactam antibiotics. For drugs whose
efficacy may be related to their peak level, for example, fluoroquinolone
antibiotics, prolongation of the dosing interval is recommended.
Related Topics
