Conclusion
| Home | | Pharmaceutical Technology |Chapter: Pharmaceutical Engineering: Conclusion
A conclusion would have added little to enlighten the reader.
Conclusion
In
the previous edition of this volume a conclusion was not presented. Even for
the novice to pharmaceutical process engineering the technical and practical
review presented was structured in a self-explanatory manner and represented
the summary of thinking over many decades in chemical and mechanical
engineering. A conclusion would have added little to enlighten the reader.
This
edition is intended to suggest that while many of the engineering principles
described previously have not changed and, indeed, are as relevant as they have
ever been, the control of the quality of the product in a demonstrable and
scientifically sound fashion has become a substantial consideration in the way
that processes are managed. The Quality by Design (QbD) principles espoused by
regulatory bodies, unlike the fundamental engineering consid-erations, are evolving
and are based on relatively new developments in the fields of multivariate
statistics and risk management. The danger in introducing these novel concepts
to an introduction to engineering principles is to dilute the clarity and
well-defined nature of the former with the more general essence of the latter.
However, it would be irresponsible to suggest, particularly to those coming to
this topic for the first time or with limited background, that engi-neering
principles alone are of relevance to pharmaceutical process engineering at this
juncture. There can be no question that the most substantial future
developments will be in new methods of analysis or data collection that can be
applied through QbD strategies involving information management and sta-tistical
assessment to deliver rapid solutions to processing problems and in all
probability to give real-time control of the variations in product output by
manipulation of input variables.
It
would be difficult to do justice to the principles of QbD, statistical
exper-imental design, and Process Analytical Technology (PAT) without expanding
the present text to a series of volumes on pharmaceutical process engineering.
However, by including them as overviews, sufficient attention is given to these
topics to give the novice a framework from which to continue to evolve an
understanding, as their importance in product development activities increases.
In
concluding this volume, a model is proposed for the relationship between the
various components described and their application to process develop-ment.
Figure 20.1 depicts the unit operations, shown originally in Figure 20.1 of
chapter 1, and indicates the role that well-designed experiments followed by
monitoring and control strategies may play in assuring the quality of the
product and, thereby, ensuring both the safety and efficacy of dosage forms
released for use in disease therapy.
The
future of pharmaceutical process engineering will relate closely to
developments in material science, analytical and information technology. It is
anticipated that many new developments, particularly with respect to
bio-technology, will be driven by efficiency in resource utilization, time, and
expense to address the medical needs of more narrowly defined patient
pop-ulations, as pharmacogenomics and the principles of individualized dosing
begin to drive requirements for smaller but more controlled production than
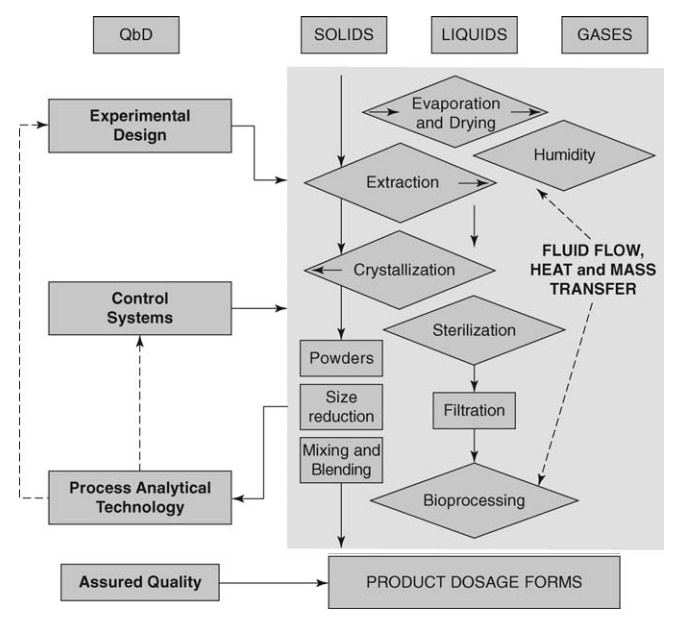
FIGURE 20.1 Monitoring and control of
elements of the manufacturing process relating to product quality.
Indeed, while it may be many years before it is a common
strategy, it can be anticipated that following statistically designed
optimization, continuous processes under direct feedback monitoring and
con-trol could routinely be used to produce product on any required scale (for
example, gram to many kilogram quantities) to supply the demand without the
need for serious depletion or accumulation of stock based on the arbitrary
scale of batch production. Linking production to demand based on well
controlled and predictable manufacturing represents an efficient commercial
strategy.
Related Topics
