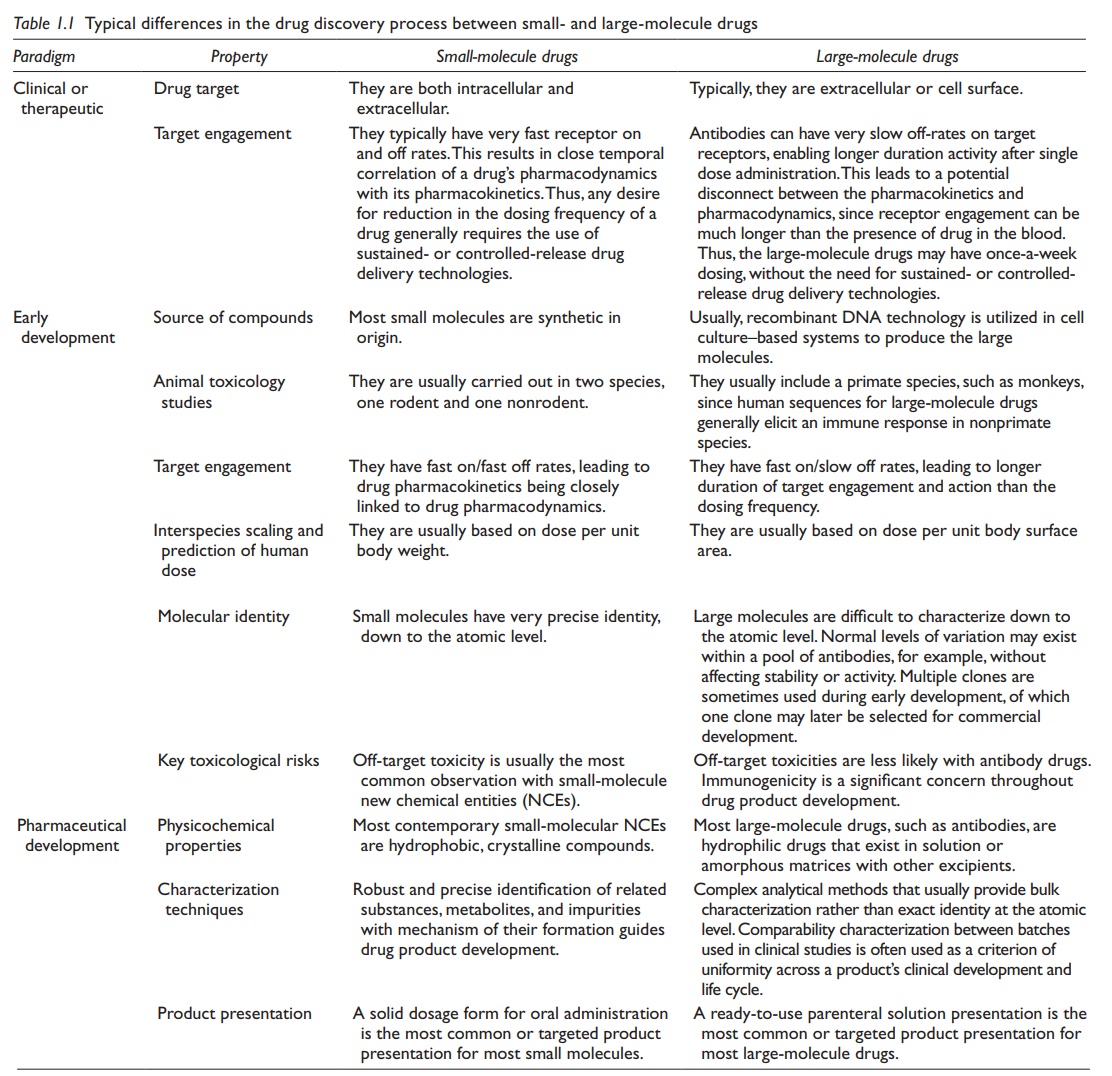
Small - versus large-molecule paradigms
| Home | | Pharmaceutical Drugs and Dosage | | Pharmaceutical Industrial Management |Chapter: Pharmaceutical Drugs and Dosage: Drug discovery
Drugs are typically divided into small and large in the contemporary vernacular based on their molecular size.
SMALL - VERSUS
LARGE-MOLECULE PARADIGMS
Drugs
are typically divided into small and large in the contemporary vernacular based
on their molecular size. The small-molecule drugs are typically the low
molecular weight compounds, such as acetaminophen and ibuprofen, with molecular
weight typically being less than or equal to 400 Da. The large-molecule drugs
are typically the high molecular weight compounds, such as antibodies and
antibody–drug conjugates, with molecular weight typically being greater than
about 4,000. This classification is based on the significantly different
molecular, biophar-maceutical, and pharmacokinetic characteristics of compounds
that fall in either of the two categories. In addition, the access to
biological targets usually differs significantly between these two broad drug
classes, with large hydrophilic antibody drugs predominantly confined to
extracellular or cell surface receptors, while the small hydrophobic drugs
being able to permeate through cell membranes and access intracellular targets.
The drugs that fall between these two broad classes in terms of molecular size
and properties are typically defined and developed on a case-by-case basis,
since their properties can be a unique mix, with dominance of one or the other
characteristic.
Although
the overall requirements and goals for new drug development are similar between
the small- and large-molecule drug candidates, there are significant
differences in the technical depth and detail as well as the pathway for these
drug candidates. For the purpose of a general compari-son, a typical
small-molecule drug is exemplified by a hydrophobic crystal-line compound
intended for oral administration, whereas a large-molecule drug is exemplified
by a monoclonal human antibody intended for paren-teral administration. These
differences are summarized in Table 1.1 and are
briefly outlined below.
Table 1.1 Typical differences in the drug
discovery process between small- and large-molecule drugs
Preclinical discovery and toxicology assessment
Large-molecule
drugs are typically produced in cell culture systems by utilizing the
recombinant DNA technology, while most small-molecule drugs are synthetic
compounds produced by using one of the laboratory high-throughput manufacturing
technologies. The synthetic chemistry utilized for generation of small-molecule
drugs are based on the principles of organic chemistry and reaction kinetics.
The utilization of cell culture for biologic manufacturing is based on
increasing scale of cell culture in bioreactors in what is termed the upstream
manufacturing operations, while the downstream manufacturing operations focus
on purifying the target protein of interest (harvesting) from the cells (Figure 1.5).
The
preclinical toxicology assessment of antibody therapeutics is typi-cally
carried out in primates to ensure relative tolerance to human sequences and
closeness of animal physiology with the human physiology that is being
targeted. In addition, more than one clone of the antibody may be tested in
animal species in certain cases in early stages of development. In the case of
small molecules, very precise crystalline form of a highly pure compound is
usually tested in the animal species to allow scaling of the observations to
human administration.
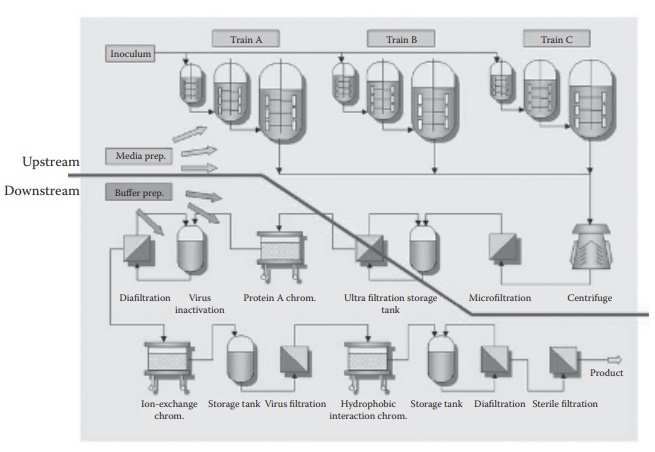
Figure 1.5 A typical process train for a
biologic-manufacturing operation showing cell culture in progressively
increasing tank sizes and cell culture volumes (upstream), and purification of
the target protein from the cultured cells (downstream) through a series of
operations yielding the target protein of interest. (From http://www.mdpi.com/2306-5354/1/4/188/htm.)
Pharmaceutical development
Small-
and large-molecule drugs are inherently different in that the typical small
molecules are hydrophobic, crystalline, well-characterized com-pounds, whereas
the typical large molecules are hydrophilic species that exist in solution. A
particular batch of an antibody may not be exactly uniform in all the molecules
that exist in solution. Antibody product pre-sentations are usually a solution,
whereas preferred product presentations for small molecules are typically in
some solid-state dosage form intended for oral administration. The ready-to-use
solution presentations of anti-bodies are typically intended for either direct
parenteral administration or dilution in intravenous fluids before
administration. In certain cases, for ensuring maximum product stability,
antibodies may be lyophilized in the presence of excipients to present a solid
dosage form in a sterile vial for reconstitution before administration.
Clinical development
Target
engagement, on and off rates, and frequency of administration vary
significantly between antibodies and small-molecule drugs. Small-molecule drugs
typically have fast on/fast off rates of target receptor occupation. This
results in their target activity being closely linked to the pharmacoki-netics
or the drug concentration at the site of action. However, for the anti-body
therapeutics, the molecular target off rates are longer. This result in
bioactivity being observed long after the drug has cleared the plasma. This can
result in longer duration of action and less frequent need for repeat
administration of the drug.
Related Topics
