Drug discovery
| Home | | Pharmaceutical Drugs and Dosage | | Pharmaceutical Industrial Management |Chapter: Pharmaceutical Drugs and Dosage: Drug discovery
Throughout history, new drug discovery has enabled human well-being and advancement, not only by ensuring survival in the wake of infections and debilitating diseases but also by steadily advancing the quality of life.
Drug discovery
Introduction
Throughout
history, new drug discovery has enabled human well-being and advancement, not
only by ensuring survival in the wake of infections and debilitating diseases
but also by steadily advancing the quality of life. For example, the discovery
of penicillin by Alexander Fleming in 1928 saved countless lives and paved the
way for the antibiotic medicines. More recent examples of life-saving medicines
include the discovery and commercializa-tion of statins for the management of
hypercholesterolemia and humanized monoclonal antibodies targeting the immune
checkpoints for the immuno-therapy of cancer. Prominent examples among the
quality-of-life improving therapeutics are the drugs for the management of
pain, hypertension, gas-trointestinal disorders, and countless others. New drug
discovery remains a continuing priority and exhibits relentless effort of
governments, private corporations, and academia alike. This chapter will
outline contemporary practices in new drug discovery and development, with an
emphasis on the interdisciplinary process, stage-gate paradigm, and the key
features that ensure safety and efficacy of every new drug through the earliest
stages of drug discovery by commercialization.
Elements of drug discovery
Early
discovery starts with the identification of disease area and molecular targets
that may be useful in the clinical setting to modulate a particular disease
condition. Several drug candidates that may be able to produce the desired
outcome at the chosen drug targets are produced. The activity of these
candidates is assessed in in vitro
assays, and candidates presenting high efficacy are shortlisted.
Structure–activity relationships (SARs) are developed to help narrow down the
drug candidates to the ones with the greatest potential for efficacy at the
lowest dose.
Early
discovery is involved not only in the generation of drug can-didates but also
in the assessment of their activity and toxicity in cell culture–based, in vitro, and in vivo systems (animal species). These often require development
of disease models on which compounds can be tested. Assessment of relative
activity and potential of different com-pounds must be made under a multitude
of criteria, thus requiring an effort to optimize
the relative performance of a compound in several attributes that are important
for success in the overall discovery and development process.
Typical
drug discovery and development process (Figure 1.1)
involves a series of iterative work streams that develop and mature in
parallel. Target selection and validation often precede first discovery efforts
and continue to
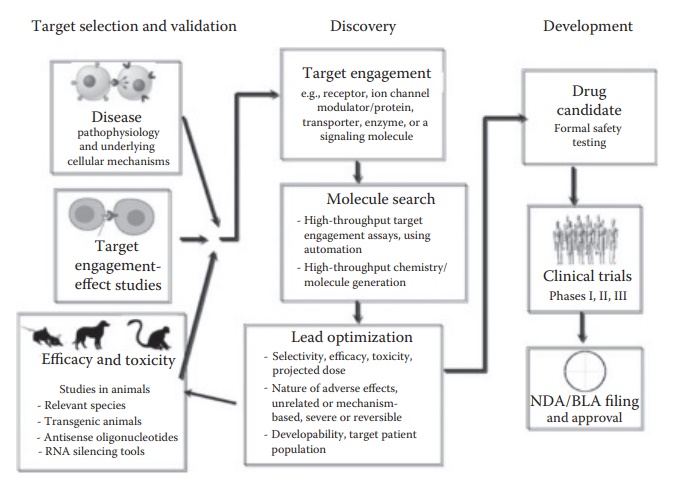
Figure 1.1 Key activities involved in drug discovery and development research.
·
Understanding disease pathophysiology
and underlying cellular or subcellular mechanisms.
·
Target
engagement–effect studies in cell culture or in
vitro systems, with a view to
develop a high-throughput assay that can be used for the screening of new drug
candidates.
·
Efficacy, toxicity, and preliminary
pharmacokinetic studies in ani-mals relevant to disease pathophysiology and
candidate drugs. These studies may involve the use of transgenic animals that
exhibit the dis-ease or modulation of particular biochemical processes and the
use of antisense oligonucelotides or RNA-silencing approaches to modulate
subcellular genetic pathways.
Discovery of new drug
candidates relies on target screening and validation efforts to not only identify the right target but also provide a
high-throughput screening assay to shortlist compounds from vast institutional
libraries and synthetic capabilities. Drug discovery efforts focus on the
following:
·
Target engagement
and evaluation of efficacy with a particular target identity and quantitative evaluate drug binding to target proteins
in cells and tissues. Quantitation of target engagement can be in terms of
percentage. The ability of a quantitated extent of interaction to pro-duce the
desired efficacy in animal models is assessed.
·
Molecule search is an
interdisciplinary involvement of quantita-tive SAR (QSAR) assessment; in silico molecular modeling studies
that try to assess molecular mechanism and location of engage-ment on the
target, which can help guide QSAR efforts; and synthe-sis/generation of new
molecules that can be tested based on these assessments.
·
Once one or more series of molecules have been identified
with key structural features needed for target engagement and efficacy, lead optimization
efforts focus on developability assessment (identifica-tion of any
liabilities that can hinder development of the compound during later stages in
the development) and appropriate modification of molecular design or delivery
strategy design to enable the progres-sion of a molecule through later stages
of drug development. These assessments also include dose projections, identification
of appropri-ate route of drug administration, and an assessment of the observed
or predictable liabilities of adverse effects.
The
drug development studies follow the
identification of a single lead molecule or drug candidate. These studies are
aimed at formal toxicity assessment that can enable initiation of
first-in-human (FIH) studies and at enabling clinical trials through various
phases of drug development into generating the needed data package for the
regulatory approval and com-mercialization. The details of the drug development
studies will be a subject of the Chapter 2.
Sources of drugs
Identification
of new molecules with the potential to produce a desired therapeutic effect
involves a combination of (1) molecular physiology and pathophysiology; that
is, research on the molecular mechanisms of biologi-cal process and disease
progression; (2) review of known therapeutic agents; and/or (3)
conceptualization and synthesis/procurement of potential new molecules that may
also involve random selection and broad biological screening.
The
sources of new drugs are varied (Figure 1.2).
NMEs can be of synthetic or natural origin, the latter involving inorganic
compounds or compounds purified from plants or animals.
Plant sources
Natural
compounds extracted from plants have often provided novel structures for
therapeutic applications. For example, vincristine is derived from the
periwinkle plant Vinca rosea,
etoposide is from the mandrake plant Podophyllum
peltatum, taxol is from the pacific yew Taxus
brevi-folia, doxorubicin is a fermentation product of the bacteria Streptomyces, l-asparaginase is from Escherichia
coli or E. cartovora, rhizoxin is
from the fungus Rhizopus chinensis,
cytarabine is from the marine sponge Cryptotethya
crypta, and bryostatin is from the sea moss Bugula neritina. Another
example is paclitaxel (Taxol®), prepared from the extract of the pacific yew,
used in the treatment of ovarian cancer.
Digoxin is one of

Figure 1.2 Different sources of drug molecules.
the
most widely used drugs in the management of congestive heart failure, weakened
heart, and irregular heart beat (arrhythmia). The common garden plant, the
foxglove or Digitalis purpurea, is
the source of digoxin.
Organic synthesis
Chemical
synthesis could involve (a) synthesis of analogs of natural compounds in an
effort to improve affinity, specificity, or potency to improve the safety and
efficacy profile of the original natural compound; (b) synthesis of a natu-ral
molecule from a more abundantly available intermediate to reduce cost and/or
improve purity (e.g., taxotere was developed to overcome the supply problems
with taxol); or (c) synthesis of a new, unique chemical structure.
Synthesis
of analogs of natural compounds is exemplified by the following: carboplatin—an
analog of cisplatin with reduced renal toxicity, doxorubicin—an analog of
daunomycin with lower cardiotoxicity, and topotecan—an analog of camptothecin
with lower toxicity. Synthesis of analogs of known drugs is sometimes aimed at
improving the targeting and the pharmacokinetics of a drug. The tauromustine
couples a nitrosourea anticancer agent to a brain-targeting peptide. Synthesis
of new molecular entities (NMEs) that are analogs of known compounds or
completely novel structures involves computer modeling of drug–receptor
interactions, followed by synthesis and evaluation by using tools such as
solid-state and combinato-rial chemistry. For example, methotrexate and
5-fluorouracil were developed as analogs of natural compounds that demonstrated
anticancer activity.
Use of animals
The
use of animals in the production of various biologic products, includ-ing
serum, antibiotics, and vaccines, has life-saving significance. Hormonal
substances, such as thyroid extract, insulin, and pituitary hormones obtained
from the endocrine glands of cattle, sheep and swine, are life-saving drugs
used daily as replacement therapy.
Genetic engineering
In
addition to the use of whole animals, cultures of cells and tissues from animal
and human origin are routinely used for the discovery and devel-opment of new
drugs—both small molecules and biologicals, such as vaccines. Drugs that were
traditionally produced in animals are increas-ingly being synthesized by using
cell and tissue cultures. The two basic technologies that drive the genetic
field of drug development are recombi-nant DNA technology and monoclonal
antibody production. Recombinant DNA technology involves the manipulation of
cellular DNA to produce desired proteins, which may then be extracted from cell
cultures for thera-peutic use. Recombinant DNA technology has the potential to
produce a wide variety of proteins. For example, human insulin, human growth
hormone, hepatitis B vaccine, and interferon are produced by recombinant DNA
technology.
A
growing class of biologics is monoclonal
antibodies against cellu-lar targets aimed for destruction, such as
molecular markers on tumors. Monoclonal antibodies target a single epitope, an
antigen surface recog-nized by the antibody, as against natural polyclonal
antibodies, which bind to different epitopes on one or more antigen molecules.
This confers a high degree of specificity to monoclonal antibodies. While
recombinant DNA techniques usually involve protein production within cells of
lower animals, monoclonal antibodies are produced in cells of higher animals,
sometimes in the patient, to ensure the lack of patient immune reaction against
these macromolecules on administration. Monoclonal antibodies are used as
anti-cancer therapeutics, in home pregnancy testing products, and for drug
tar-geting to specific sites within the body. In home pregnancy testing
products, the monoclonal antibody used is highly sensitive to binding at one
site of the human chorionic gonadotropin (HCG) molecule, a specific marker to
preg-nancy because HCG is synthesized exclusively by the placenta.
Gene therapy
Gene
therapy is the process of correction or replacement of defective genes and has
the potential to be used to prevent, treat, cure, diagnose, or miti-gate human
disease caused by genetic disorders. Oligonucleotides and small interfering RNA
(siRNA) are used to inhibit aberrant protein production, whereas gene therapy
aims at expressing therapeutic proteins inside the body.
Related Topics
