Structure of Amino Acids
| Home | | Biochemistry |Chapter: Biochemistry : Amino Acids
Although more than 300 different amino acids have been described in nature, only 20 are commonly found as constituents of mammalian proteins.
STRUCTURE
Although more than 300
different amino acids have been described in nature, only 20 are commonly found
as constituents of mammalian proteins. [Note: These are the only amino acids
that are coded for by DNA, the genetic material in the cell.] Each amino acid
has a carboxyl group, a primary amino group (except for proline, which has a
secondary amino group), and a distinctive side chain (“R group”) bonded to the
α-carbon atom (Figure 1.1A). At physiologic pH (approximately 7.4), the
carboxyl group is dissociated, forming the negatively charged carboxylate ion
(–COO–), and the amino group is protonated (–NH3+). In
proteins, almost all of these carboxyl and amino groups are combined through
peptide linkage and, in general, are not available for chemical reaction except
for hydrogen bond formation (Figure 1.1B). Thus, it is the nature of the side
chains that ultimately dictates the role an amino acid plays in a protein. It
is, therefore, useful to classify the amino acids according to the properties
of their side chains, that is, whether they are nonpolar (have an even
distribution of electrons) or polar (have an uneven distribution of electrons,
such as acids and bases) as shown in Figures 1.2 and 1.3.
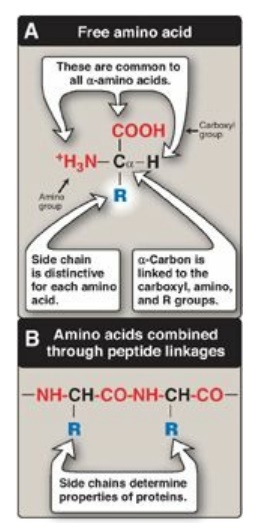
Figure 1.1 Structural features of amino acids (shown in their fully protonated form).
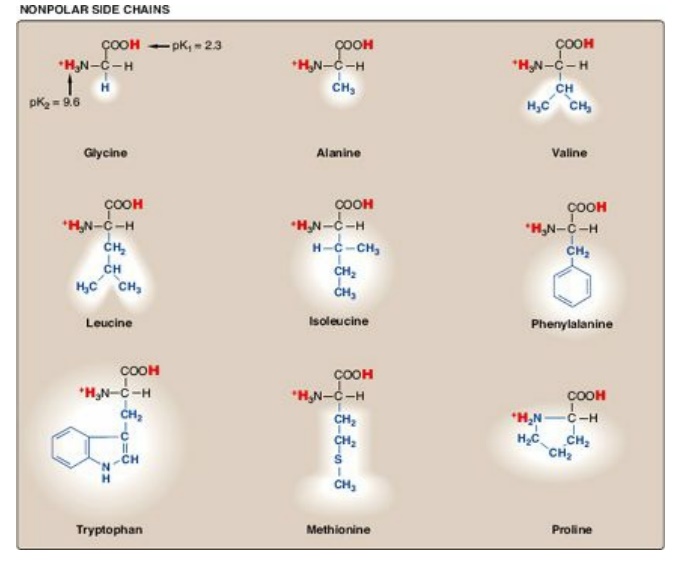
Figure 1.2 Classification of the 20 amino acids commonly found in proteins, according to the charge and polarity of their side chains at acidic pH is shown here and continues in Figure 1.3. Each amino acid is shown in its fully protonated form, with dissociable hydrogen ions represented in red print. The pK values for the α-carboxyl and α-amino groups of the nonpolar amino acids are similar to those shown for glycine.
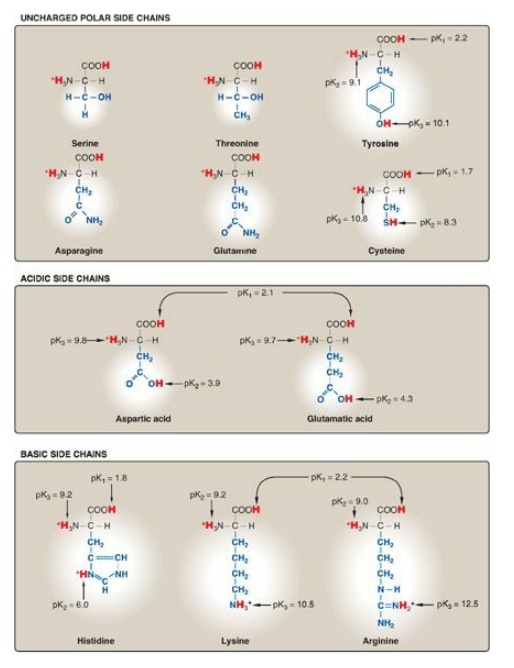
Figure 1.3 Classification of the 20 amino acids commonly found in proteins, according to the charge and polarity of their side chains at acidic pH (continued from Figure 1.2).
A. Amino acids with nonpolar side chains
Each of these amino
acids has a nonpolar side chain that does not gain or lose protons or
participate in hydrogen or ionic bonds (see Figure 1.2). The side chains of these
amino acids can be thought of as “oily” or lipid-like, a property that promotes
hydrophobic inter-actions (see Figure 2.10).
1. Location of nonpolar amino acids in proteins: In proteins found in aqueous
solutions (a polar environment) the side chains of the nonpolar amino acids
tend to cluster together in the interior of the protein (Figure 1.4). This
phenomenon, known as the hydrophobic effect, is the result of the
hydrophobicity of the nonpolar R groups, which act much like droplets of oil
that coalesce in an aqueous environment. The nonpolar R groups, thus, fill up
the interior of the folded protein and help give it its three-dimensional
shape. However, for proteins that are located in a hydrophobic environment,
such as a membrane, the nonpolar R groups are found on the outside surface of
the protein, interacting with the lipid environment see Figure 1.4. The
importance of these hydrophobic interactions in stabilizing protein structure.
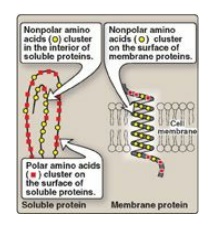
Figure 1.4 Location
of nonpolar amino acids in soluble and membrane proteins.
Sickle cell anemia, a
sickling disease of red blood cells, results from the replacement of polar glutamate
with nonpolar valine at the sixth position in the β subunit of hemoglobin.
2. Proline: Proline differs from other amino acids in that its
side chain and α-amino N form a rigid, five-membered ring structure (Figure
1.5). Proline, then, has a secondary (rather than a primary) amino group. It is
frequently referred to as an “imino acid.” The unique geometry of proline
contributes to the formation of the fibrous structure of collagen and often
interrupts the α-helices found in globular proteins.
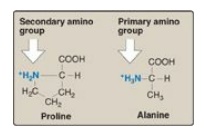
Figure 1.5 Comparison of the secondary amino group found in proline with the primary amino group found in other amino acids such as alanine.
B. Amino acids with uncharged polar side chains
These amino acids have
zero net charge at physiologic pH, although the side chains of cysteine and
tyrosine can lose a proton at an alkaline pH (see Figure 1.3). Serine,
threonine, and tyrosine each contain a polar hydroxyl group that can
participate in hydrogen bond formation (Figure 1.6). The side chains of
asparagine and glutamine each contain a carbonyl group and an amide group, both
of which can also participate in hydrogen bonds.
1. Disulfide bond: The side chain of cysteine contains
a sulfhydryl (thiol) group (– SH), which is an important component of the
active site of many enzymes. In proteins, the –SH groups of two cysteines can
be oxidized to form a covalent cross-link called a disulfide bond (–S–S–). Two
disulfide-linked cysteines are referred to as “cystine.”
Many extracellular
proteins are stabilized by disulfide bonds. Albumin, a blood protein that
functions as a transporter for a variety of molecules, is an example.
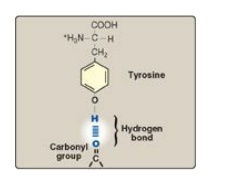
Figure 1.6
Hydrogen bond between the phenolic hydroxyl group of tyrosine and another
molecule containing a carbonyl group.
2. Side chains as sites of attachment for other
compounds: The
polar hydroxyl group of serine; threonine; and, rarely, tyrosine, can serve as
a site of attachment for structures such as a phosphate group. In addition, the
amide group of asparagine, as well as the hydroxyl group of serine or
threonine, can serve as a site of attachment for oligosaccharide chains in
glycoproteins.
C. Amino acids with acidic side chains
The amino acids
aspartic and glutamic acid are proton donors. At physiologic pH, the side
chains of these amino acids are fully ionized, containing a negatively charged
carboxylate group (–COO–). They are, therefore, called aspartate or glutamate
to emphasize that these amino acids are negatively charged at physiologic pH
(see Figure 1.3).
D. Amino acids with basic side chains
The side chains of the basic amino acids accept protons (see Figure 1.3). At physiologic pH, the R groups of lysine and arginine are fully ionized and positively charged. In contrast, histidine is weakly basic, and the free amino acid is largely uncharged at physiologic pH. However, when histidine is incorporated into a protein, its R group can be either positively charged (protonated) or neutral, depending on the ionic environment provided by the protein. This is an important property of histidine that contributes to the buffering role it plays in the functioning of proteins such as hemoglobin. [Note: Histidine is the only amino acid with a side chain that can ionize within the physiologic pH range.]
E. Abbreviations and symbols for commonly occurring amino acids
Each amino acid name
has an associated three-letter abbreviation and a one-letter symbol (Figure
1.7). The one-letter codes are determined by the following rules.

Figure 1.7 Abbreviations and symbols for the commonly occurring amino acids.
1. Unique first letter: If only one amino acid begins with
a given letter, then that letter is used as its symbol. For example, V =
valine.
2. Most commonly occurring amino acids have
priority: If
more than one amino acid begins with a particular letter, the most common of
these amino acids receives this letter as its symbol. For example, glycine is
more common than glutamate, so G = glycine.
3. Similar sounding names: Some one-letter symbols sound like
the amino acid they represent. For example, F = phenylalanine, or W =
tryptophan (“twyptophan” as Elmer Fudd would say).
4. Letter close to initial letter: For the remaining amino acids, a
one-letter symbol is assigned that is as close in the alphabet as possible to
the initial letter of the amino acid, for example, K = lysine. Furthermore, B
is assigned to Asx, signifying either aspartic acid or asparagine, Z is
assigned to Glx, signifying either glutamic acid or glutamine, and X is
assigned to an unidentified amino acid.
F. Optical properties of amino acids
The α-carbon of an
amino acid is attached to four different chemical groups (asymmetric) and is,
therefore, a chiral, or optically active carbon atom. Glycine is the exception
because its α-carbon has two hydrogen substituents. Amino acids with an asymmetric
center at the α-carbon can exist in two forms, designated D and L, that are
mirror images of each other (Figure 1.8). The two forms in each pair are termed
stereoisomers, optical isomers, or enantiomers. All amino acids found in
proteins are of the L configuration. However, D-amino acids are found in some
antibiotics and in bacterial cell walls.
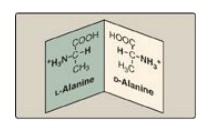
Figure 1.8 D and L forms of alanine are mirror images.
Related Topics
