Bioactivation and Tissue Toxicity
| Home | | Biopharmaceutics and Pharmacokinetics |Chapter: Biopharmaceutics and Pharmacokinetics : Biotransformation of Drugs
Formation of highly reactive metabolites (from relatively inert chemical compounds) which interact with the tissues to precipitate one or more of the several forms of toxicities such as carcinogenesis and teratogenesis is called as bioactivation or toxicological activation.
BIOACTIVATION AND TISSUE TOXICITY
Formation of highly reactive metabolites (from relatively inert chemical
compounds) which interact with the tissues to precipitate one or more of the
several forms of toxicities such as carcinogenesis and teratogenesis is called
as bioactivation or toxicological activation. The
reactive, chemically unstable species, capable of toxication, are broadly
divided into two categories (see Fig. 5.4.) —
·
Electrophiles
·
Free radicals.
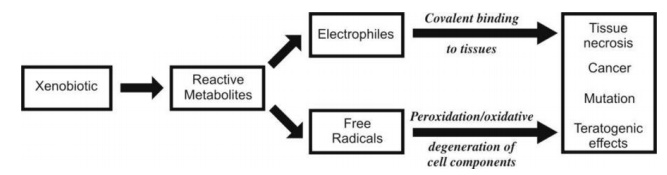
Fig. 5.4. Mechanisms of tissue toxicity by
bioactivation of drugs
Electrophiles are species deficient in electron pair. The
enzyme system through which they are
generated is cytochrome P-450. Carbon, nitrogen or sulphur-containing compounds
can be metabolically activated to yield electrophiles. Important electrophiles
are epoxides (e.g. epoxide of benzo(a)pyrene present in cigarette smoke which
causes cancer), hydroxylamines, nitroso and azoxy derivatives, nitrenium ions
and elemental sulphur. The mechanism by which electrophiles precipitate
toxicity is through covalent binding to nucleophilic tissue components such as
macromolecules (proteins, nucleic acids and lipids) or low molecular weight
cellular constituents. Covalent binding to DNA is responsible for
carcinogenicity and tumour formation. The body’s defence against electrophiles
is their inactivation by conjugation with glutathione, the most abundant
cellular nucleophile with -SH group. An example of tissue toxicity due to
electrophiles is hepatotoxicity of paracetamol metabolites.
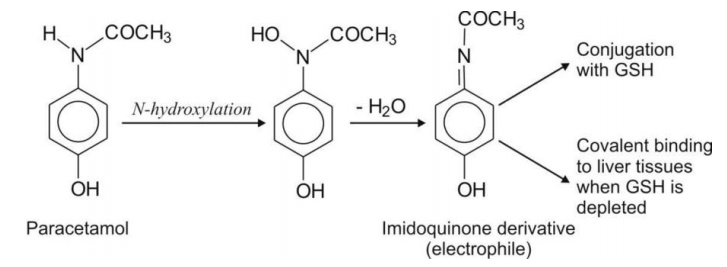
Free Radicals: are species containing an odd number of electrons. They may be positively
charged (cation radical), negatively
charged (anion radical) or neutral (neutral radical).

Free radicals are generally formed via NADPH cytochrome P-450 reductase or
other flavin containing reductases. Xenobiotics that on metabolic activation
yield free radicals are quinones, arylamines, nitroaryls and carbon
tetrachloride. Endogenous compounds such as epinephrine and DOPA can also
generate free radicals. Most free radicals are organic. They produce toxicity
by peroxidation of cellular components. An important class of free radicals is
inorganic free radicals such as hydrogen peroxide (H2O2)
and superoxide anion (O2-).
These oxidative moieties can cause tremendous
tissue damage leading to mutations or cancer. The potential toxicity of free
radicals is far greater than that of the electrophiles. Cellular defence
mechanisms against free radicals include control imposed by membrane structure,
neutralization by glutathione, control exerted by non-enzymatic antioxidant
scavengers such as vitamins A, E and C and enzymatic inactivation of oxygen
derived free radicals.
Generation of reactive metabolites is indicated by
modification in enzyme activities, formation of glutathione conjugates (or
mercapturic acids) and depletion in tissue levels of glutathione. Since the
availability of glutathione in the body determine the threshold for toxic
response, thiols (e.g. N-acetyl cysteine) can be used to treat poisoning by
drugs such as paracetamol that yield reactive metabolites.
Table 5.7 lists some of the compounds whose
metabolites are tissue reactive.
TABLE 5.7
Compounds and their Metabolic Reaction that Generate Toxic Intermediates

Related Topics
