Hydrolytic Reactions
| Home | | Biopharmaceutics and Pharmacokinetics |Chapter: Biopharmaceutics and Pharmacokinetics : Biotransformation of Drugs
These reactions differ from oxidative and reductive reactions in 3 respects:
PHASE I REACTIONS
HYDROLYTIC REACTIONS
These reactions differ
from oxidative and reductive reactions in 3 respects:
1. The reaction does not involve
change in the state of oxidation of the substrate.
2. The reaction results in a
large chemical change in the substrate brought about by loss of relatively
large fragments of the molecule.
3. The hydrolytic enzymes that
metabolise xenobiotics are the ones that also act on endogenous substrates.
Moreover, their activity is not confined to liver as they are found in many
other organs like kidney, intestine, etc.
A number of functional groups are hydrolysed viz. esters, ethers, amides, hydrazides,
etc.
Hydrolysis of Esters and Ethers
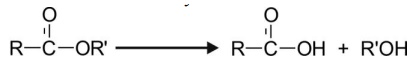
Esters on hydrolysis yield alcohol and carboxylic
acid. The reaction is catalysed by esterases.
The ester substrates undergoing hydrolysis can be classified as under:
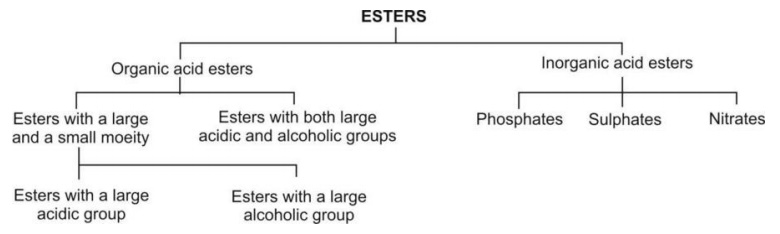
Organic esters with both large acidic and alcoholic
groups on hydrolysis results in metabolites with complete loss of activity.
Esters where one of the groups is relatively large, retain much of their
activity when hydrolysed since such a group is generally a pharmacophore (having
pharmacological activity). In many cases, such esters are prodrugs which rely on hydrolysis for their
transformation into active form, e.g. chloramphenicol palmitate.
Aromatic esters are hydrolysed by arylesterases and
aliphatic esters by carboxylesterases.
Examples of various classes of esters undergoing
hydrolysis are given below.
Organic acid (carboxylic acid) esters
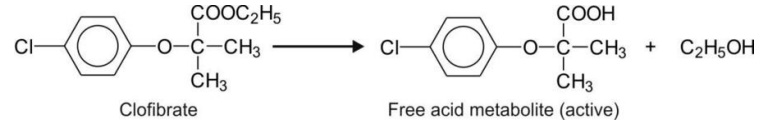
Esters with a large acidic (and small alcohol) group e.g. clofibrate.
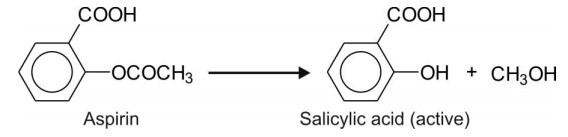
Esters with large alcoholic (and small acidic) group e.g. aspirin.
Esters with large acidic and alcoholic
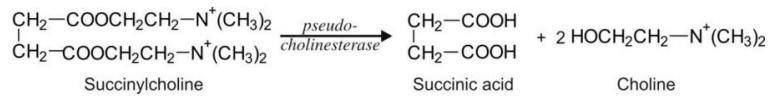
groups (generally amine alcohols) e.g. succinylcholine.
Inorganic Acid Esters
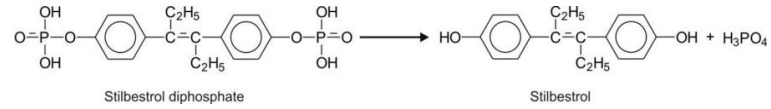
Phosphates e.g. stilbestrol diphosphate.
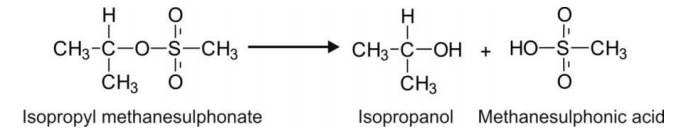
Sulphates e.g. isopropyl methanesulphonate.
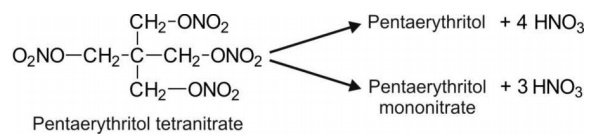
Nitrates e.g. pentaerythritol tetranitrate
Ethers undergoing hydrolysis are glycosides such as
digoxin and digitoxin and O-glucuronides.
Hydrolysis of Amides (C-N bond cleavage)
Amides are hydrolysed slowly in comparison to
esters. The reaction, catalysed by amidases,
involves C-N cleavage to yield carboxylic acid and amine.
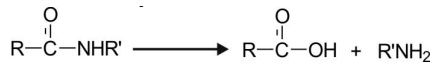
Primary amides are rare. Secondary
amides form the largest group of amide drugs.
Examples of amide hydrolysis are given below.
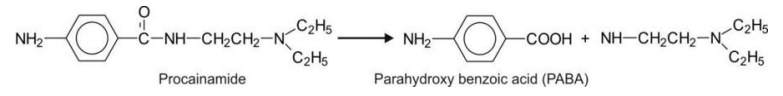
Secondary amides with aliphatic substituent on N-atom e.g. procainamide (hydrolysed slowly
in comparison to procaine)
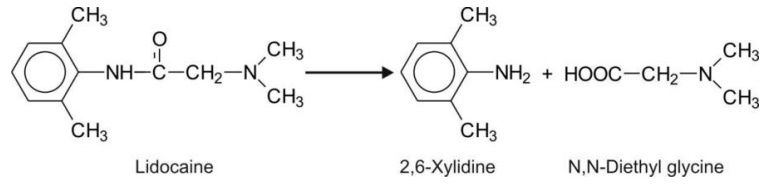
Secondary amides with aromatic substituent on N-atom (anilides) e.g. lidocaine.
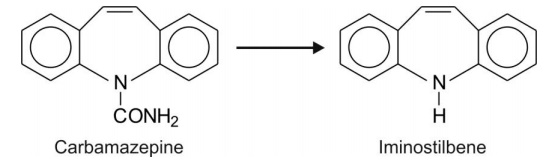
Tertiary amides (N-atom contained in a ring) e.g. carbamazepine.
Hydrazides are also a class of amides e.g.
isocarboxazide.
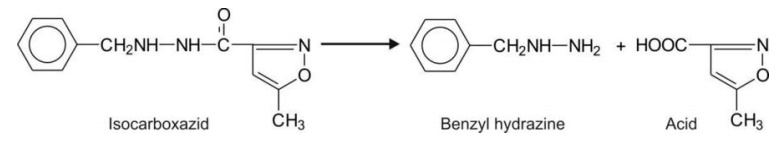
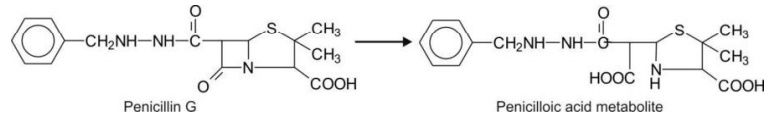
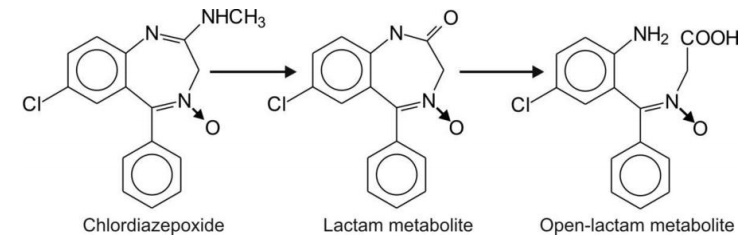
Hydrolytic Cleavage of Non-aromatic Heterocycles
Nonaromatic heterocycles also contain amide
functions, e.g. lactams (cyclic amides). Several lactams that undergo
hydrolysis are:
1. Four-membered lactams (ß-lactam) e.g.
penicillins.
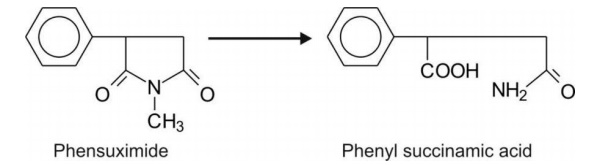
2. Five-membered lactams e.g.
succinimides.
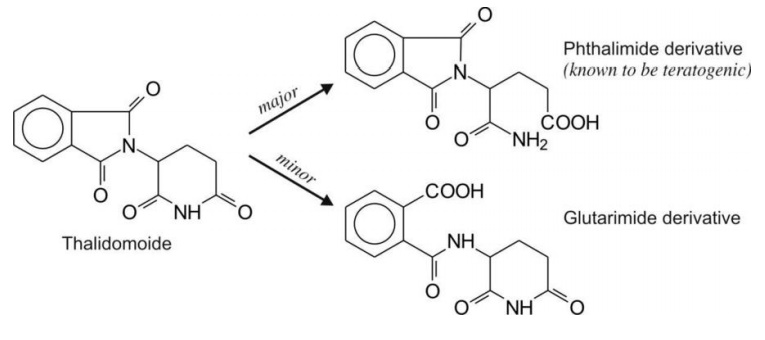
3. Six-membered lactams e.g.
thalidomide.
4. Seven-membered lactams e.g.
chlordiazepoxide
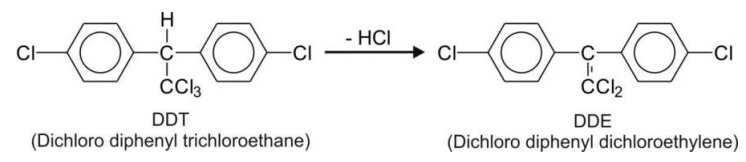
Hydrolytic Dehalogenation
Chlorine atoms attached to aliphatic carbons are dehalogenated easily, e.g. DDT.
Miscellaneous Hydrolytic Reactions
These reactions include hydration of epoxides and
arene oxides, hydrolysis of sulphonyl ureas, carbamates, hydroxamates and of
glucuronide and sulphate conjugates.
Related Topics
