Chapter Summary, Study Questions
| Home | | Biochemistry |Chapter: Biochemistry : Structure of Proteins
Central to understanding protein structure is the concept of the native conformation, which is the functional, fully folded protein structure (for example, an active enzyme or structural protein).
CHAPTER SUMMARY
Central to
understanding protein structure is the concept of the native conformation
(Figure 2.15), which is the functional, fully folded protein structure (for
example, an active enzyme or structural protein). The unique three-dimensional
structure of the native conformation is determined by its primary structure,
that is, its amino acid sequence. Interactions between the amino acid side
chains guide the folding of the polypeptide chain to form secondary, tertiary,
and (sometimes) quaternary structures, which cooperate in stabilizing the
native conformation of the protein. In addition, a specialized group of
proteins named chaperones is required for the proper folding of many species of
proteins. Protein denaturation results in the unfolding and disorganization of
the protein’s structure, which are not accompanied by hydrolysis of peptide
bonds. Denaturation may be reversible or, more commonly, irreversible. Disease
can occur when an apparently normal protein assumes a conformation that is
cytotoxic, as in the case of Alzheimer disease and t h e transmissible
spongiform encephalopathies (TSEs), including Creutzfeldt-Jakob disease. In
Alzheimer disease, normal proteins, after abnormal chemical processing, take on
a unique conformational state that leads to the formation of neurotoxic amyloid
β peptide (Aβ) assemblies consisting of β-pleated sheets. In TSEs, the
infective agent is an altered version of a normal prion protein that acts as a
“template” for converting normal protein to the pathogenic conformation.
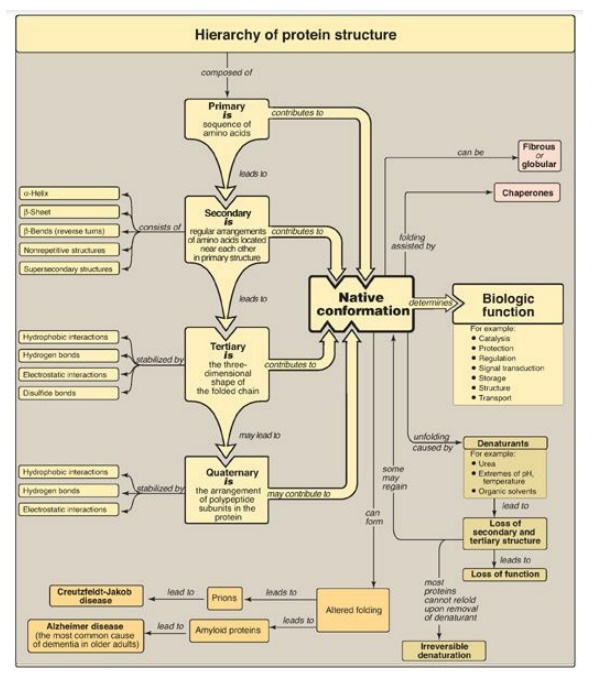
Figure 2.15
Key concept map for protein structure.
Study Questions
Choose the ONE best answer.
2.1 Which one of the following statements
concerning protein structure is correct?
A. Proteins consisting
of one polypeptide have quaternary structure that is stabilized by covalent
bonds.
B. The peptide bonds
that link amino acids in a protein most commonly occur in the cis
configuration.
C. The formation of a
disulfide bond in a protein requires the participating cysteine residues to be
adjacent in the primary structure.
D. The denaturation of
proteins leads to irreversible loss of secondary structural elements such as
the α-helix.
E. The primary driving force for protein folding is
the hydrophobic effect.
Correct answer = E. The hydrophobic effect, or the
tendency of nonpolar entities to associate in a polar environment, is the
driving force of protein folding. Quaternary structure requires more than one
polypeptide, and, when present, it is stabilized primarily by noncovalent
bonds. The peptide bond is almost always trans. The two cysteine residues
participating in disulfide bond formation may be a great distance apart in the
amino acid sequence of a polypeptide (or on two separate polypeptides) but are
brought into close proximity by the three-dimensional folding of the
polypeptide. Denaturation may be reversible or irreversible.
2.2 A particular point mutation results in
disruption of the α-helical structure in a segment of the mutant protein. The
most likely change in the primary structure of the mutant protein is:
A. glutamate to
aspartate.
B. lysine to arginine.
C. methionine to
proline.
D. valine to alanine.
Correct answer = C. Proline, because of its secondary
amino group, is incompatible with an α-helix. Glutamate, aspartate, lysine, and
arginine are charged amino acids, and valine is a branched amino acid. Charged
and branched (bulky) amino acids may disrupt an α-helix.
2.3 In comparing the α-helix to the β-sheet, which
statement is correct only for the β-sheet?
A. Extensive hydrogen
bonds between the carbonyl oxygen (C=O) and the amide hydrogen (N-H) of the
peptide bond are formed.
B. It may be found in
typical globular proteins.
C. It is stabilized by interchain hydrogen bonds.
D. it is an example of
secondary structure.
E. It may be found in
supersecondary structures.
Correct answer = C. The β-sheet is stabilized by
interchain hydrogen bonds formed between separate polypeptide chains and by
intrachain hydrogen bonds formed between regions of a single polypeptide. The
α-helix, however, is stabilized only by intrachain hydrogen bonds. Statements
A, B, D, and E are true for both of these secondary structural elements.
2.4 An 80-year-old man presented with impairment of
higher intellectual function and alterations in mood and behavior. His family
reported progressive disorientation and memory loss over the last 6 months.
There is no family history of dementia. The patient was tentatively diagnosed
with Alzheimer disease. Which one of the following best describes Alzheimer
disease?
A. It is associated
with β-amyloid, an abnormal protein with an altered amino acid sequence.
B. It results from
accumulation of denatured proteins that have random conformations.
C. It is associated
with the accumulation of amyloid precursor protein.
D. It is associated with the deposition of
neurotoxic amyloid β peptide aggregates. E. It is an environmentally produced
disease not influenced by the genetics of the individual.
F. It is caused by the
infectious β-sheet form of a host-cell protein.
Correct answer = D. Alzheimer disease is associated
with long, fibrillar protein assemblies consisting of β-pleated sheets found in
the brain and elsewhere. The disease is associated with abnormal processing of
a normal protein. The accumulated altered protein occurs in a β-pleated sheet
configuration that is neurotoxic. The amyloid β that is deposited in the brain
in Alzheimer disease is derived by proteolytic cleavages from the larger
amyloid precursor protein, a single transmembrane protein expressed on the cell
surface in the brain and other tissues. Most cases of Alzheimer disease are
sporadic, although at least 5% of cases are familial. Prion diseases, such as
Creutzfeldt-Jakob, are caused by the infectious β-sheet form (PrPSc ) of a
host-cell protein (PrPc).
Related Topics
