Synthesis of Glycosaminoglycans
| Home | | Biochemistry |Chapter: Biochemistry : Glycosaminoglycans, Proteoglycans, and Glycoproteins
The heteropolysaccharide chains are elongated by the sequential addition of alternating acidic and amino sugars donated by their uridine diphosphate (UDP)-derivatives.
SYNTHESIS OF GLYCOSAMINOGLYCANS
The
heteropolysaccharide chains are elongated by the sequential addition of alternating
acidic and amino sugars donated by their uridine diphosphate (UDP)-derivatives.
The reactions are catalyzed by a family of specific glycosyltransferases. The
synthesis of GAGs is analogous to that of glycogen except that GAGs are
produced for export from the cell. Their synthesis occurs, therefore, primarily
in the Golgi, rather than in the cytosol.
A. Synthesis of amino sugars
Amino sugars are
essential components of GAGs, glycoproteins, and glycolipids and are also found
in some antibiotics. The synthetic pathway of amino sugars is very active in
connective tissues, where as much as 20% of glucose flows through this pathway.
1. N-Acetylglucosamine and N-acetylgalactosamine: The monosaccharide fructose
6-phosphate is the precursor of N-acetyl-glucosamine (GlcNAc),
N-acetylgalactosamine (GalNAc), and the sialic acids, including
N-acetylneuraminic acid (NANA). In each of these sugars, a hydroxyl group of
the precursor is replaced by an amino group donated by glutamine (Figure 14.8).
[Note: The amino groups are then almost always acetylated.] The UDP-derivatives
of GlcNAc and GalNAc are synthesized by reactions analogous to those described
for UDP-glucose synthesis. These nucleotide sugars are the activated forms of
the monosaccharides that can be used to elongate the carbohydrate chains.
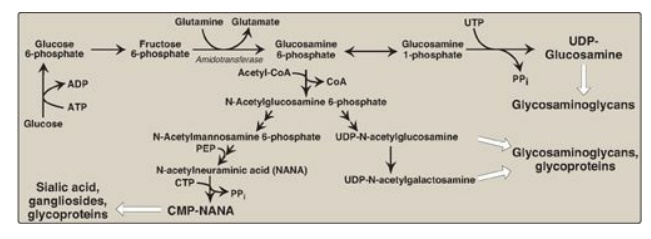
Figure 14.8 Synthesis of the amino sugars. UTP = uridine triphosphate; UDP = uridine diphosphate; CoA = coenzyme A; PEP = phosphoenoylpyruvate; CTP = cytidine triphosphate; CMP = cytidine monophosphate; PPi = pyrophosphate.
2. N-Acetylneuraminic acid: NANA, a nine-carbon, acidic monosaccharide, is a member of the family of sialic acids, each of which is acylated at a different site. These compounds are usually found as terminal carbohydrate residues of oligosaccharide side chains of glycoproteins; glycolipids; or, less frequently, of GAGs. The carbons and nitrogens in NANA come from N-acetylmannosamine and phosphoenolpyruvate (an intermediate in the glycolytic pathway;). [Note: Before NANA can be added to a growing oligosaccharide, it must be converted into its active form by reacting with cytidine triphosphate (CTP). The enzyme CMP-NANA synthetase catalyzes the reaction. This is the only nucleotide sugar in human metabolism in which the carrier nucleotide is a monophosphate.]
B. Synthesis of acidic sugars
D-Glucuronic acid,
whose structure is that of glucose with an oxidized carbon 6 (– CH2OH
→ –COOH) and its C-5 epimer, L-iduronic acid, are essential components of GAGs.
Glucuronic acid is also required in detoxification reactions of a number of
insoluble compounds, such as bilirubin, steroids, and many drugs, including the
statins. In plants and mammals (other than guinea pigs and primates, including
humans), glucuronic acid serves as a precursor of ascorbic acid (vitamin C).
The uronic acid pathway also provides a mechanism by which dietary D-xylulose
can enter the central metabolic pathways.
1. Glucuronic acid: Glucuronic acid can be obtained in
small amounts from the diet. It can also be obtained from the lysosomal
degradation of GAGs, or from glucose 6-phosphate via the uronic acid pathway.
The end product of glucuronic acid metabolism in humans is D-xylulose 5-phosphate,
which can enter the pentose phosphate pathway and produce the glycolytic
intermediates glyceraldehyde 3-phosphate and fructose 6-phosphate (Figure 14.9;
see also Figure 13.2). The active form of glucuronic acid that donates the
sugar in GAG synthesis and other glucuronidation reactions is UDP-glucuronic acid,
which is produced by oxidation of UDP-glucose (Figure 14.10).
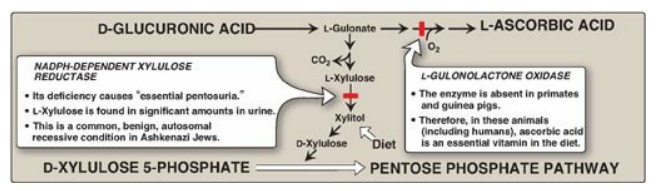
Figure 14.9 Metabolism of glucuronic acid. NADPH = reduced nicotinamide adenine dinucleotide phosphate.
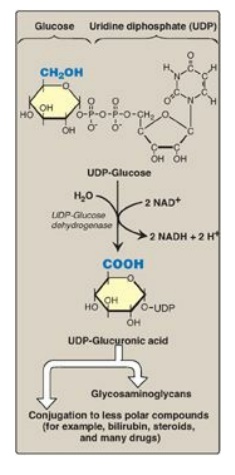
Figure 14.10 Oxidation of UDP-glucose to UDPglucuronic acid. NAD(H) = nicotinamide adenine dinucleotide.
2. L-Iduronic acid synthesis: Synthesis of L-iduronic acid
residues occurs after D-glucuronic acid has been incorporated into the carbohydrate
chain. Uronosyl 5-epimerase causes epimerization of the D- to the L-sugar.
C. Synthesis of the core protein
The core protein is synthesized by ribosomes on the rough endoplasmic reticulum (RER) and enters the RER lumen. The protein is then glycosylated by membrane-bound glycosyltransferases located in the Golgi.
D. Synthesis of the carbohydrate chain
Carbohydrate chain
formation begins by synthesis of a short linkage region on the core protein on
which carbohydrate chain synthesis will be initiated. The most common linkage
region is formed by the transfer of a xylose from UDP-xylose to the hydroxyl
group of a serine (or threonine) catalyzed by xylosyltransferase. Two galactose
molecules are then added, completing the trihexoside. This is followed by
sequential addition of alternating acidic and amino sugars (Figure 14.11) and
epimerization of some D-glucuronyl to L-iduronyl residues.
E. Addition of sulfate groups
Sulfation of a GAG
occurs after the particular monosaccharide to be sulfated has been incorporated
into the growing carbohydrate chain. The source of the sulfate is
3’-phosphoadenosyl-5’-phosphosulfate ([PAPS], a molecule of adenosine
monophosphate with a sulfate group attached to the 5’-phosphate). The sulfation
reaction is catalyzed by sulfotransferases. [Note: An example of the synthesis
of a sulfated GAG, chondroitin sulfate, is shown in Figure 14.11.] PAPS is also
the sulfur donor in glycosphingolipid synthesis.
A defect in the sulfation of the growing
glycosaminoglycan chains results in one of several autosomal recessive
disorders, the chondrodystrophies, that affect the proper development and
maintenance of the skeletal system.
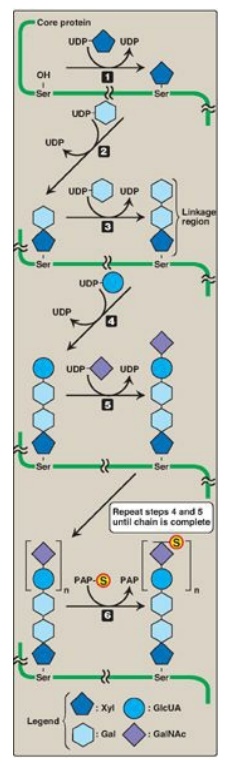
Figure 14.11 Synthesis of
chondroitin sulfate.
Related Topics
