Pharmacovigilance
Pharmacovigilance - Covers all topics with images and clear explanation. Study Pharmaceutical Microbiology Online course, study material, important questions and answers, Notes, textbook.

PART I: THE BASIS OF PHARMACOVIGILANCE
Chapter 1: The Basis of Pharmacovigilance
=> The Basis of Pharmacovigilance
=> Diagnosing Adverse Drug Reactions
=> Current Methods of Pharmacovigilance
=> Hypothesis-Generating Methods
=> Hypothesis-Testing Methods
=> Conclusion
Chapter 2: Legal Basis - EU
=> Legal Basis � EU
=> Guidance
=> Definitions
=> European Pharmacovigilance for Medicinal Products
=> European Pharmacovigilance for Centrally
=> European Pharmacovigilance Legislation
Chapter 3: Legal Basis - United States
=> Legal Basis - United States
=> Background
=> Law
=> Regulations
=> Food and Drug Administration Reporting Standards For Investigational Drugs and Biological Products
=> Food and Drug Administration Reporting Standards for Marketed Drugs and Biological Products
=> Reporting Adverse Product Experiences from Marketed Products
=> Guidance Documents
=> Enforcement
=> Conclusion
Chapter 4: Ethical Oversight, Consent and Confidentiality
=> Ethical Oversight, Consent and Confidentiality
=> Confidentiality Issues in Epidemiology Studies
=> Existing Law
=> State Laws
=> Research Ethics
=> Implications and Next Steps
Chapter 5: Pharmacovigilance-Related Topics at the Level of the International Conference on Harmonisation
=> Pharmacovigilance-Related Topics at the Level of the International Conference on Harmonisation
=> The ICH Step Process
=> The Pharmacovigilance-Related ICH Topics
=> Discussion and Conclusions
Chapter 6: Periodic Safety Update Reports
=> Periodic Safety Update Reports
=> Purpose of the PSUR
=> PSUR - General Principles
=> Regulatory Requirements
=> PSUR Content
=> Summary Bridging Reports
=> Addendum Reports
=> The PSUR Process
=> Best Methods of Compliance
=> Pragmatic Solutions
=> Clinical Trial Annual Reports
=> PSUR and Risk Management
=> Conclusion
Chapter 7: Non-Clinical Safety Evaluation and Adverse Events in Phase I Trials
=> Non-Clinical Safety Evaluation and Adverse Events in Phase I Trials
=> Non-Clinical Testing Requirements
=> Use of Animals to Predict Human Toxicities
=> Estimation of Safe Starting Dose and Safety Assessment and Risk�Benefit Analysis
=> Adverse Events in Clinical Trials
=> Conclusions
Chapter 8: Mechanisms of Adverse Drug Reactions
=> Mechanisms of Adverse Drug Reactions
=> Importance of Adverse Drug Reactions
=> Classification of Adverse Drug Reactions
=> Type a Adverse Drug Reactions
=> Type B or Idiosyncratic Adverse Drug Reactions
=> The Role of Drug Metabolism in Type B Adverse Drug Reactions
=> An Example of a Drug That Causes Toxicity Through the Formation of a Chemically Reactive Intermediate
=> The Role of the Immune System in Type B Adverse Drug Reactions
=> The Role of Viruses in Type B Adverse Drug Reactions
=> Genetic Predisposition to Type B Adverse Drug Reactions
=> Conclusion
Chapter 9: Micturin and Torsades de Pointes
=> Micturin and Torsades de Pointes
Chapter 10: Withdrawal of Terodiline: A Tale of Two Toxicities
=> Withdrawal of Terodiline: A Tale of Two Toxicities
=> Drug-Induced QT Interval Prolongation and Pharmacovigilance Planning (ICH E2E)
=> Re-Birth of Terodiline
=> Terodiline-Induced Proarrhythmias
=> Limitations of Formal Post-Marketing Surveillance Studies
=> Initial Regulatory Deliberations
=> Similarities Between Terodiline and Prenylamine
=> Prenylamine-Induced Proarrhythmias
=> Polymorphic CYP2D6-Mediated Stereoselective Metabolism
=> Pharmacokinetics and Recommended Dose Schedules
=> Pharmacodynamic Similarity to Prenylamine
=> Stereoselectivity in Proarrhythmic Potential
=> Lessons to be Learnt
=> Why the Regulatory Concerns on Drug-Induced QT Interval Prolongation?
=> Electrophysiological Basis of Torsade De Pointes
=> Drug-Induced QT Interval Prolongation and Regulatory Guidance
=> Preclinical Investigations of the �QT-Liability� of a Drug
=> Pre-Approval Clinical Safety Dataset
=> Exceptional Circumstances Requiring Extended Database
=> Risk�Benefit Assessment
=> Development of Single Enantiomers or Metabolites of Marketed Racemic Drugs
Chapter 11: Nomifensine and Haemolytic Anaemia
=> Nomifensine and Haemolytic Anaemia
=> Background
=> Haemolytic Anaemia
=> Post-Marketing Experience 1977�82
=> Post-Marketing Experience 1983�86
=> Discussion
PART II: SIGNAL GENERATION
Chapter 12: WHO Programme - Global Monitoring
=> History of WHO Programme - Global Monitoring
=> Present Programme Structure
=> Current Work of WHO
=> What is Still Missing � What We Must Do in the Future
=> Joining the WHO Programme
=> Conclusions
Chapter 13: Medical Dictionary for Regulatory Activities (MedDRA)
=> Medical Dictionary for Regulatory Activities (MedDRA�)
=> Uses of MedDRA
=> MedDRA Structure
=> MedDRA Rules and Conventions
=> Using MedDRA for Data Entry
=> Database Searches and Data Retrieval
=> Data Analysis and Presentation
=> MedDRA and Labelling
Chapter 14: Regulatory Pharmacovigilance in the EU
=> Introduction and Historical Perspective
=> Legal Basis, Principles and Organisation of the EU Pharmacovigilance System
=> The Process of Regulatory Pharmacovigilance in the EU
=> Future Challenges and Conclusions
Chapter 15: Spontaneous Reporting - UK
=> Spontaneous Reporting - UK
=> Background
=> Purpose and Achievements of the Yellow Card Scheme
=> Weaknesses of Yellow Cards
=> Reporting Volumes
=> Recent Initiatives to Enhance the Scheme
=> Widening the Yellow Card Reporting Base
=> Specialist Therapeutic Areas
=> Suspected ADRS in Children
=> Unlicensed Herbal Remedies
=> Facilitation of Reporting - New Technology and Media
=> The Anonymised Yellow Card
=> Independent Review of Access to the Yellow Card Scheme
=> Focus on Patients
=> Future Directions for the Yellow Card Scheme
Chapter 16: Spontaneous Reporting - France
=> The French Pharmacovigilance System
=> History and Organisation
=> Regional Pharmacovigilance Centres
=> Source and Management of Reports
=> Alert Management
=> Results
=> The French Imputability Method
=> Basic Principles
=> Time Sequence Analysis
=> Signs and Symptoms
=> Future Perspectives
Chapter 17: Spontaneous Reporting in Germany
=> Spontaneous Reporting in Germany
=> Demographic and Economic Data
=> Licences for Medicinal Products in Germany
=> Actors in Spontaneous Reporting
=> Adverse Drug Reaction Databases
=> Identifying Safety Issues From the Spontaneous Reporting System
=> Risk-Assessment Procedures
=> Risk Communication
=> Conclusions
Chapter 18: Spontaneous Reporting - United States
=> Spontaneous Reporting - United States
=> Adverse Drug Reaction Reporting in the United States
=> Adverse Drug Reaction Reports Review Practices at the Food and Drug Administration
=> Methods of Signal Detection and Refinement
=> Case Series
=> Regulatory Action Based on Spontaneous Reports
=> The Value and Future of Pharmacovigilance in the United States
Chapter 19: Statistical Methods of Signal Detection
=> Statistical Methods of Signal Detection
=> What Constitutes a Signal?
=> Classification of ADR Reports
=> Characteristics of Spontaneous Reports
=> Proportional Reporting Ratios
=> Rationale for Proportional Methods
=> The Use of PRRs in Monitoring Drugs
=> Further Developments and Key Issues
=> Conclusions
Chapter 20: Statistical Methods of Evaluating Pharmacovigilance Data
=> Statistical Methods of Evaluating Pharmacovigilance Data
=> Risk Estimaton From SR
Chapter 21: Data Mining in Pharmacovigilance
=> Data Mining in Pharmacovigilance: A View from the Uppsala Monitoring Centre
=> Data Mining
=> Description of Data Mining Methodology Used by the Uppsala Monitoring Centre
=> �Validation� of the Data Mining Approach
=> Comparison of Methods
=> The Limitations and Use of Data Mining
Chapter 22: Pharmacovigilance in the Netherlands
=> History and Organization
=> The Netherlands Pharmacovigilance Centre Lareb
=> Further Initiatives in Pharmacovigilance in the Netherlands
Chapter 23: CIOMS Working Groups and their Contribution to Pharmacovigilance
=> CIOMS Working Groups and their Contribution to Pharmacovigilance
=> CIOMS I - Expedited Reporting of Individual ADRs
=> CIOMS IA - Harmonisation of Data Elements and Fields for Electronic Reporting of Individual ADRs
=> CIOMS II - Periodic Safety Updates
=> CIOMS III - Core Clinical Safety Information
=> CIOMS IV - Benefit�Risk Evaluation
=> CIOMS V Good Case Management and Reporting Practices
=> CIOMS VI - Management Of Safety Information From Clinical Trials
=> CIOMS VII - The Future
=> Conclusion
Chapter 24: PEM in the UK
=> Background
=> Method
=> Results
=> Discussion
=> Conclusion
Chapter 25: PEM in New Zealand
=> PEM in New Zealand
=> Background
=> Selection of Medicines for Monitoring
=> OverView of Intensive Medicines Monitoring Programme Methodology
=> Prescription Data
=> Identification of Events
=> Processing of Events
=> Privacy and Ethical Considerations
=> Outputs of PEM in New Zealand
=> Routine Analysis of Data
=> Specific Studies Using Intensive Medicines Monitoring Programme Data
=> Conclusions
Chapter 26: MEMO in the United Kingdom
=> MEMO in the United Kingdom
=> Description of the Database
=> Prescription Drug Data
=> Hospital Data
=> Other in-Hospital and Outcome Data Sets
=> Record-Linkage of Other Databases
=> Current Areas of Investigation
=> Confidentiality and Ethics in Memo
=> Strengths
=> Weaknesses
=> Future Developments in Memo and Conclusion
Chapter 27: The General Practice Research Database
=> The General Practice Research Database: Now and the Future
=> Historical Overview
=> Characteristics of General Practice Research Database 2005
=> Future Developments in the UK National Health Service
=> Current and Future Developments in General Practice Research Database
=> Strengths and Weaknesses of the General Practice Research Database
=> Conclusion
Chapter 28: Overview of North American Databases
=> Overview of North American Databases
=> Group Health Cooperative of Puget Sound
=> Kaiser Permanente Medical Care Program
=> Kaiser Permanente Northwest
=> Unitedhealth Group
=> Medicaid Databases
=> Health Databases in Saskatchewan
=> HMO Research Network
=> Vaccine Safety Datalink: A Special Purpose Database
=> Weighing In
Chapter 29: Other Databases in Europe for the Analytic Evaluation of Drug Effects
=> Other Databases in Europe for the Analytic Evaluation of Drug Effects
=> The Netherlands
=> Computerization of the Dutch Community Pharmacy System
=> Denmark
=> Italy
Chapter 30: Surveillance for Medical Devices - USA
=> Surveillance for Medical Devices - USA
=> Premarket Overview
=> Postmarket Surveillance Context
=> The Future
=> Addendum
Chapter 31: Pharmacovigilance and Risk Management in Japan
=> Pharmacovigilance and Risk Management in Japan
=> Spontaneous Reporting System (SRS) in Japan
=> Amendment of Pharmaceutical Affairs Law (PAL) in 2002
=> Enhancement of Post-Marketing Safety Measures
=> Good Vigilance Practice (GVP) And ADR Reports Via Companies
=> Good Postmarketing Study Practice (GPSP) and ICH E2E Guideline
=> The New Type of Investigations Using Pharmacoepidemiologic Methods in Japan
=> Novel Trend of Pharmacovigilance and Risk Management by the Regulatory Body
=> Role of Academia and Non-Governmental Bodies in the Future Pharmacovigilance in Japan
=> Risk Management in Japan
PART III: PHARMACOVIGILANCE AND SELECTED SYSTEM ORGAN CLASSES
Chapter 32: Dermatological ADRs
=> Dermatological ADRs
=> Patterns of Cutaneous ADRS
=> Other Drug-Induced Cutaneous Reactions
=> Assessment and Reporting of Cutaneous ADRS
Chapter 33: Gastrointestinal ADRs
=> Gastrointestinal ADRs
=> Non-Steroidal Anti-Inflammatory Drugs
=> Bisphosphonates
=> Selective Serotonin Re-Uptake Inhibitors
=> Pancreatic Enzyme Supplements
=> Rotavirus Vaccine
=> Measles-Mumps�Rubella Vaccine
=> Haematological ADRs
=> Mechanisms of ADR-Causing Cytopenias
=> Mechanisms of ADR-Affecting Haemostasis
=> Some Examples of Individual ADR - Pharmacovigilance in Action
=> Management of Haematological ADR
=> Strategies for the Detection and Prevention of Idiosyncratic Haematological ADRS
=> Conclusion
Chapter 35: Hepatic ADRs
=> Hepatic ADRs
=> Definitions
=> Epidemiology
=> Mechanisms of Drug-Induced Liver Injury
=> Management of Idiosyncratic Hepatotoxicity
Chapter 36: Ocular Side Effects of Prescription Medications
=> Ocular Side Effects of Prescription Medications
=> Hydroxychloroquine (Plaquenil)
=> Isotretinoin (Accutane)
=> Sildenafil (Viagra)
=> Corticosteroids � Inhaled
=> Tamoxifen (Nolvadex)
=> Amiodarone (Cordarone)
=> Topiramate (Topamax)
=> Bisphoshonates: Pamidronate Disodium, Alendronic Acid, Risedronate Sodium Etidronate Disodium and Olpadronate
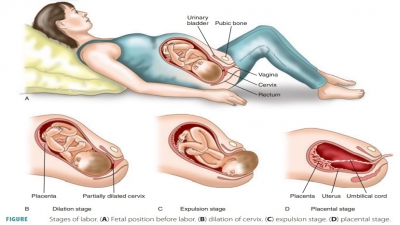
Chapter 37: Drug Safety in Pregnancy
=> Drug Safety in Pregnancy
=> Frequency and Variety of Medication Use Among Pregnant Women
=> Pre-Marketing Sources of Data Regarding Reproductive and Developmental Safety of Prenatal Drug Exposures
=> Post-Marketing Sources of Data Regarding Reproductive and Developmental Safety of Prenatal Drug Exposures
=> Monitoring for Pregnancy Exposures and Pregnancy Prevention for Known Human Teratogens
=> Challenges for the Future
Chapter 38: Renal Adverse Drug Reactions
=> Renal Adverse Drug Reactions
=> Definitions and Diagnosis
=> Epidemiology
=> Mechanisms of Renal Adverse Drug Reactions
=> Diagnosis of Renal Adverse Drug Reactions
=> Prevention of Renal Adverse Drug Reactions
=> Conclusion
Chapter 39: Anaesthetic Adverse Drug Reactions
=> Anaesthetic Adverse Drug Reactions
=> Historical Perspectives
=> Anaphylactic Reactions
=> Individual Agents
=> Specific Problems
Chapter 40: Pharmacovigilance in Pediatrics
=> Background and Introduction to Pediatric Issues
=> Why Pediatric Studies and a Special Focus on Safety Reporting are Necessary?
=> Experiences From Specific Focused Pediatric Postmarketing Review
=> Why the Pediatric Population May Be More Vulnerable
=> Data Sources for Postmarketing Pediatric Adverse Drug Events
=> Safety Signal Detection and Evaluation in the Pediatric Population
=> Challenges for Surveillance of Drug Adverse Events
Chapter 41: The Cardiovascular Spectrum of Adverse Drug Reactions
=> The Cardiovascular Spectrum of Adverse Drug Reactions
=> Dofetilide
=> Appetite Suppressants
=> Oestrogen and Venous Thromboembolism
Chapter 42: Drugs and the Elderly
=> The Ageing Population and Changing Demography
=> Disease Prevalence and Drug Use in the Elderly
=> Interactions in Relation to Multiple Drug Prescribing
=> Altered Pharmacokinetics in the Elderly
=> Altered Pharmacodynamics in the Elderly
=> ADRS in the Elderly and Definition of an ADR
=> Incidence of ADRS in the Elderly
=> Implications for Drug Development and Use
PART IV: KEY CURRENT TOPICS
Chapter 43: US Activities in Risk Management of Pharmaceutical Products
=> US Activities in Risk Management of Pharmaceutical Products
=> Risk Management Guidances
=> Appendix: Guidance for Industry - Good Pharmacovigilance Practices and Pharmacoepidemiologic Assessment
Chapter 44: Risk Management - a European Regulatory View
=> Risk Management - a European Regulatory View
=> Background
=> European Focus on ADRS
=> Excellence in Pharmacovigilance Model
=> European Pharmaceutical Legislation
=> European Risk Management Plans
=> Safety Specification
=> Future Development of European Risk Management Strategies
=> Conclusion
Chapter 45: The Efficacy and Safety of Selective Serotonin Reuptake Inhibitors for the Treatment of Depression in Children and Adolescents
=> Abstract
=> Introduction
=> Efficacy From Published Trials
=> Meta-Analysis of Published and Unpublished Studies
=> Safety From Clinical Trial Data
=> Safety From Observational Studies
=> New Studies to Address the Research Question
=> Improving the Safety Infrastructure
=> Recent FDA Actions and Consequences
=> Conclusions
Chapter 46: Pharmacoepidemiology of Hormone Therapy: An Evolving Picture
=> Pharmacoepidemiology of Hormone Therapy: An Evolving Picture
=> Controversy Over WHI Results
=> Discussion
=> The Future
Chapter 47: NSAIDs - COX-2 Inhibitors Risks and Benefits
=> NSAIDs - COX-2 Inhibitors - Risks and Benefits
=> Post-Marketing Cardiovascular Safety Signal
=> Spontaneous Reports
=> Cardiovascular Safety Signal Evaluation
=> Large Trials of Cox-2 Inhibitors in Disease Prevention
=> Meta-Analysis of Cox-2 Inhibitors and Cardiovascular Outcomes
=> Observational Studies Published after the Withdrawal of Rofecoxib
=> Cardiovascular Safety of Other Cox-2 Inhibitors
=> Comments
Chapter 48: Introduction to Pharmionics
=> Introduction and Background of Pharmionics
=> Pharmionics in Overview
=> Methodological Issues in Compiling Drug Dosing Histories of Ambulatory Patients
=> Electronic Medication Event Monitoring
=> Case Studies
=> Lessons Learned
PART V: LESSONS AND DIRECTIONS
Chapter 49: Teaching and Learning Pharmacovigilance
=> Teaching and Learning Pharmacovigilance
=> Current Influences on Pharmacovigilance Education
=> The �Mistakes and Mishaps in Healthcare� Movement
=> �Safety� as a Construct in Healthcare
=> Public Aspirations for �Drug Safety�
=> Learning Pharmacovigilance
=> Interactive Techniques for Learning Pharmacovigilance
=> Teaching Pharmacovigilance
=> Conclusions
Chapter 49B: Practical Experience in Teaching Pharmacovigilance
=> Practical Experience in Teaching Pharmacovigilance
Chapter 50: Fatal Medication Errors and Adverse Drug Reactions - Coroners Inquests and Other Sources
=> Fatal Medication Errors and Adverse Drug Reactions - Coroners� Inquests and Other Sources
=> The Consequences of Adverse Drug Events
=> The Literature Relating to Fatal Adverse Drug Events
=> Information From Coroners� Inquests
=> Search Strategies
=> Results
=> Overall Summary of the Three Series
=> The Genesis of Medication Errors
=> The Lessons From Deaths Related to Medication
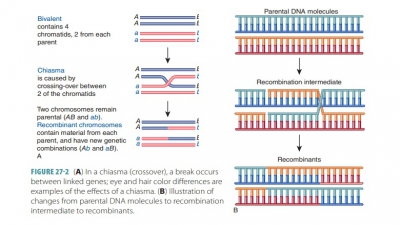
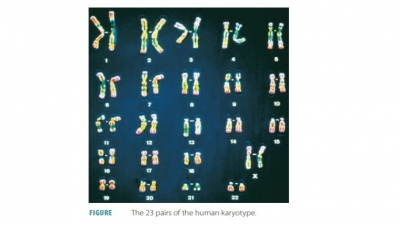
Chapter 51: Pharmacogenetics and the Genetic Basis of ADRs
=> Pharmacogenetics and the Genetic Basis of ADRs
=> Pharmacogenetics
=> The Genetic Basis of ADRS
=> The Changing Paradigm of Drug Development and Delivery
=> The Impact of Pharmacogenetics on Clinical Drug Development
=> Conclusion
Chapter 52: Keynote Clinical Lessons from Pharmacovigilance
=> Keynote Clinical Lessons from Pharmacovigilance
=> Pharmacovigilance
=> Spontaneous Reporting Schemes
=> Disease Registries
=> Follow-Up Studies
=> Multipurpose Databases
=> Evidence-Based Medicine
=> Hospital Drug Monitoring
=> Genome Research and Pharmacovigilance
=> Responses to Risks
=> Conclusions